Contents
Use of Alternative Materials in Cement Manufacturing
by Javed I. Bhatty* and John Gajda**
[wpecpp name=”package” price=”75″ align=”center”]
Cement manufacturing utilizes a variety of naturally occurring raw materials such as limestone, clay, shale, sand, iron ore, and gypsum as the main ingredients. In addition, because of chemical compatibility, today as in the past, alternative materials such as industrial wastes, by-products, and other marginal materials can be easily and economically utilized as raw material in cement manufacture. These materials are being generated in abundance as a result of growing industrialization across the globe.

Figure 2.4.1. A cement plant in North America using alternative materials in the cement raw mix.
The focus of this chapter is the use of industrial wastes and by-products in the production of cement. Owing to the compositional compatibility of several of these wastes and by-products with the raw materials used in cement manufacture, their potential for reutilization is gaining ground. Figure 2.4.1 shows a cement plant in North America using alternative materials as component of raw feed. The degree of their usefulness as a substitute raw material, however, depends primarily on economic considerations as well as the chemical composition of the alternate material or mate-rials that they replace.
In recent years, cement manufacturing has successfully made use of a number of wastes and by-products as supplementary raw materials. Typically these materials are lime-bearing carbonates, paper sludge, lime waste from water purification plants, sludge from sugar and fertilizer industries; slag from the iron, steel, and other metal industries; fly ash, bottom ash, and desulfurization sludge from power plants; red mud, mineral and ore tailings, catalyst fines, foundry sand; and sludge from sewage treatment plants. Several marginal raw materials have also been used as substitutes.
The prominent characteristics of the wastes and by-products and their compatibility with the cement raw feed, their degree of substitution, and the properties of clinker and cement produced, along with the derived material, economical, and environmental benefits to the cement industry are discussed in this chapter.
The principal raw materials in cement manufacturing are calcareous materials (containing calcium carbonates) such as limestone, chalk, marl, and marble; siliceous materials (containing silica) such as sand and sandstone; and argillaceous materials (containing alumina and silica) such as clay, shale, and slate. Other corrective materials such as bauxite and iron ore are frequently used to permit easy burnability. A carefully proportioned mixture of these materials is fired to produce clinker. Occasionally, though, fluxes and mineralizers have also been used to promote clinkering to further conserve thermal energy and improve production.
It is critical that the composition of the waste or the by-product considered for use in the cement raw mix is compatible with that of the cement raw materials they replace or supplement. Based on this criterion, a number of wastes and byproduct materials have been identified as partial or total substitutes for raw material in cement manufacture. Several marginal raw materials have also been used as substitutes.
In the following sections, the materials in question are classified according to their calcareous, siliceous, argillaceous, and other chemical characteristics. In many cases, some materials span more than one of these categories. The best example is metallurgical slag that can be rich in lime, silica, and alumina. Although the focus of this chapter is on materials that are used as kiln feed, some materials can be interground with clinker or blended with cement. The best example of this type of material is fly ash.
As a final note, this information is not intended to be inclusive of all waste materials or by-prod-ucts utilized in the manufacture of portland cement.Instead this chapter provides an overview of many utilized materials, their use, and results.
MATERIALS REPLACING CALCAREOUS COMPONENTS
Calcareous materials are rich in lime (calcium oxide). They primarily replace, partially or fully, the calcareous component (limestone) of the cement raw feed based upon their chemical composition and compatibility with the replacing materials. Examples of their use in cement manufacture are given in the following section.
Marginal Limestones
Gradual depletion of quality limestone for cement manufacture has prompted widespread use of marginal and lower grade limestone in recent years. Ahluwalia and Page (1992) have discussed an Indian experience on the use of a low-grade limestone high in magnesium and silica. They classi-fied this limestone according to the magnesia and silica contents for use in cement raw feed (see Table 2.4.1).
Table 2.4.1. Components of Marginal Limestones
In an earlier study, Ahluwalia and others (1981) beneficiated low-grade limestones prior to their use and found them suitable for cement manufacturing. Out of the several beneficiation tech-niques employed, a flotation method proved most useful, despite higher energy consumption. The beneficiation involved size reduction by grinding and separation of the impurities by flotation to obtain a concentrate with an increased calcium content. Other methods considered were differen-tial grinding and sieving, electrostatic separation, photometric sorting, and bacterial leaching. These limestones were regarded as more suitable to wet and semi-wet cement manufacturing processes, where they could be conveniently processed by flotation and used directly for raw feed preparation. Although more suitable for wet process, the upgraded limestone has also been successfully used in other cement manufacturing processes. One Indian cement plant used the beneficiated limestone in a kiln with two-stage preheater and calciner to produce 1780 tons/day clinker (Purohit and others, 1987; Shrinkhande and others, 1991). The microstructure of this clinker featured well-formed alite and belite crystals distributed in interstitial phases. The esti-mated Bogue composition of clinker was: 41% C3S, 48% C2S, 5% C4AF, and 6% C3A. The cement produced had normal strength, setting, and other physical properties.
A South African cement plant also used marginal limestones in raw mix for producing normal clinker and cements (Du Toit, 1996). In separate trials, two low-grade limestones (both high in silica) replaced 10% and 14% raw mix respectively; the balance was high-grade limestone, magnetite, and bottom ash. The composition of limestones used in the trials is shown in Table 2.4.2. The composition of the high-grade limestone used is also shown for comparison.
Table 2.4.2. Composition of Select Limestones, Mass %
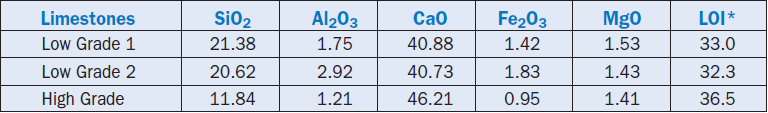
*LOI = loss on ignition
Under identical burning conditions, the trial using 10% low-grade limestone (and 80% high grade, 8.5% bottom ash, and 1% magnetite) produced clinker and cement of acceptable quality. Phase distribution of alite and belite were normal for the clinker, and the cement showed comparable physical characteristics to that of normally produced cement.
Use of low-grade limestone together with high-grade limestone “sweeteners” is now becoming common in a number of U.S. cement plants for material as well as economical reasons. The cements produced are comparable to those produced with the typical limestones.
Cement Kiln Dust (CKD)
Cement kiln dust is generated from the cement plants as fine particulate material swept from the rotary kiln and collected in a baghouse or electrostatic precipitator. Cement kiln dust (CKD) is often extremely rich in lime. Normally, all CKD that can be returned to the process is returned, except for cases where alkali considerations prohibit it. In wet kilns, it is injected through mid-kiln devices or even insufflated into the burning zone.
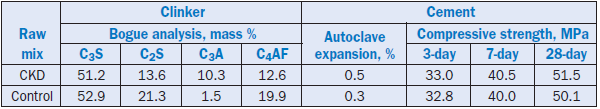
Typically, CKD is composed of partially calcined raw material, clinker particles, and alkali compounds swept out of the kiln with the exit gases. The reuse of CKD in kiln feed for vertical shaft kilns has been reported by Raina and others (1997). The analysis of dust used for demonstra-tion is given in Table 2.4.3. The use of CKD was maximized.
Table 2.4.3. Composition of CKD, Mass %

The demonstration showed that although the alkalies and chloride contents in the CKD were high, the cement displayed good strength and met the soundness criterion. Select results on clinker and cement are shown in Table 2.4.4.
Table 2.4.4. Bogue Analysis of Clinker and Physical Properties of Cement Made with CKD
It must, however, be kept in mind that since CKDs are also high in alkalies, their reuse in the raw feed has to be carefully controlled to avoid producing clinkers and cements with unacceptably high alkalies. For durability reasons, low-alkali cements are more desirable for use in concrete than high-alkali cements. For this particular reason, attempts are made to separate alkali compounds from CKDs prior to their reutilization.
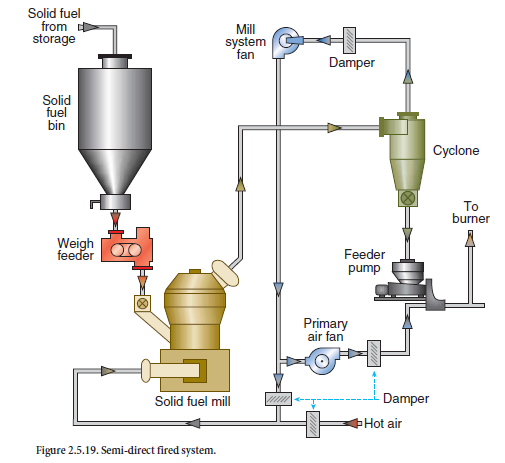
The practice of using CKD by mid-kiln injection in long wet kilns is typical. Since the addition of CKD to slurry in a wet process is counter-beneficial as it stiffens the slurry and requires high water demand to maintain slurry flow, CKD is injected at a mid-kiln location. The practice does not effect the thermal profile of the kiln as the CKD routinely becomes part of the kiln feed. Figure 2.4.2 shows the mid-kiln kiln injection of CKD in a long-wet kiln at a cement plant in the Midwest region of the United States.

Figure 2.4.2. Mid-kiln injection of cement kiln dust (CKD) in a long wet-process kiln.
Fuller Company (1992) has beneficiated high-alkali and high-sulfate CKD by using a fluidized bed process. The clinker thus produced was separated from its alkali and sulfur and interground with normal clinker to produce an ASTM C150 compatible cement. In addition, the alkali concentrate can be sold as a separate product. Young and Morrison (1991) developed Passamaquoddy scrubber technology for removing alkali compounds from CKD. The alkali compounds are separately marketed whereas the alkali-free CKD is reused as a component in cement raw feed.
Carbonate Sludge
Use of a lime-rich sludge from the fertilizer industry in the raw meal of a wet process cement plant has also been referred to by Ahluwalia and Page (1992). In addition to the lime, the sludge con-tained elevated levels of sulfate (6%), fluoride (1.2%), and phosphate (1.8%). These “impurities” restricted the use of sludge to 50% of the kiln feed. The composition of the sludge is given in Table 2.4.5.
Table 2.4.5. Composition of Carbonate Sludge from Fertilizer Plant

The sludge partially replaced the high-grade limestone already used at the plant. Excessive use of sludge affected the strength and setting properties of cement, most likely due to the presence of phosphate and fluoride. However, the addition of sludge could be adjusted to overcome this prob-lem. The burnability of the raw feed was significantly improved at reduced temperatures, primarily because of the fluxing ability of fluoride present in the sludge. Burnability of the raw feed in terms of free-lime reduction and the degree of lime-combinability in the clinkers is shown in Table 2.4.6.
Table 2.4.6. Burnability of Raw Meal with the Carbonate Sludge
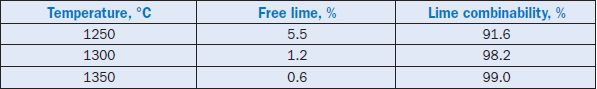
* Lime Combinability = % [(Total CaO – Free CaO)/ Total CaO]
A 10% use of phosphochalk, another lime-bearing sludge containing high contents of sulfate
(9.9%), phosphate (2.4%), and fluoride (2.0%), was also tested in a raw mix that contained lime-stone, marl, and sandstone (NCB Report, 1990). Addition of phosphochalk beyond 10% was not considered because of the high phosphate and fluoride contents. Microscopic examination of the clinker showed well-formed C3S (48%) and C2S (31%), and low free lime.
Carbide Sludge
Lime-rich carbide sludge has also been used as a partial limestone replacement in cement raw mix (NCB Report, 1987). Due to the high chloride content, and its effect on the flow properties of raw meal in a wet-process cement plant, the sludge could only be used for up to 30% of the raw feed. The addition of sludge showed easy burning of the kiln feed, possibly due to better reactivity of the sludge, and the fact that at least some of the calcium is in the form of hydroxide, which requires a lower calcination energy than CaCO3. Clinker made from the raw feed with sludge showed well-developed alite and belite crystals that produced cements with good performance characteristics.
Blast Furnace Slag
Blast furnace slag is a by-product of iron smelting operations. Both crys-tallized and vitreous (glassy) slags are produced. The crystalline slag is allowed to cool slowly after it is dis-charged from the blast furnace, while the vitreous slag is quenched with water as it is discharged from the furnace. Studies have demonstrated that both crystallized and vitreous slags are suitable supplements for cement raw feed. Figure 2.4.3. shows a slag pile produced from one of the iron and steel operations in the United States.

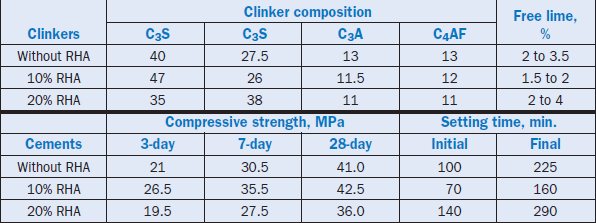
Fig. 2.4.3. Blast furnace slag generated at an iron smelting plant.
Texas Industries, Inc. (Young, 1995) patented the process of using blast furnace slag or basic oxygen furnace slag as feed stock components for cement clinker production in long rotary kilns. In their process, unground slag is added to the raw mix as it clinkers well without grinding. Up to 30% slag has been used to produce clinker of acceptable chemical and phase composition. Typical blast furnace slag used along with the processing parameters and phase composition of the resulting clinkers are shown in Table 2.4.7. The process saves a significant amount of grinding energy.
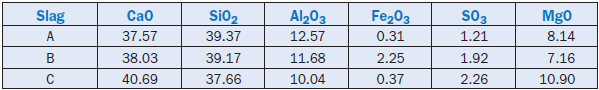
In another application, Puertas and others (1988) demonstrated the use of crystallized blast furnace slags in producing cement clinker. Being rich in CaO, SiO2, and Al2O3 (Table 2.4.8), the slags replaced considerable amounts of limestone, silica, and clay in the raw mix.
Tests were carried out using 37% ground slag in a raw mix having a lime saturation factor of 98, a silica modulus of 2.18, and an alumina modulus of 2.2. The addition of slag altered the kinetics of clinker formation. Above 1350°C the reactivity of the raw mix was good, and the clinker had normal formation and distribution of phases. Higher temperatures (1450°C) and finer slag grains (90µm) improved the burnability of the raw mix that produced clinker with normal mineralogical composition and a better distribution of clinker phases. Overall, the crystalline blast furnace slag was found more suitable than the glassy slag for cement manufacture, with a potential for improved operation and energy conservation.
Table 2.4.7. Composition of Slag, Raw Mix Parameters, and Clinker Phase Analyses
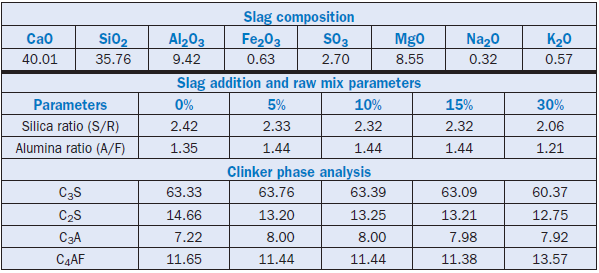
Table 2.4.8. Composition of Different Crystalline Blast Furnace Slags, Mass %
Blast Furnace Slag and Converter Slag Mixtures
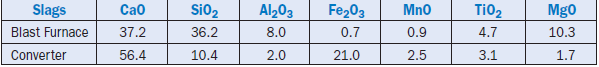
Monshi and Asgarani (1999) made a combined use of blast furnace slag and converter steel slags as raw feed components. Compositions of the slags used are shown in Table 2.4.9.
Table 2.4.9. Composition of Blast Furnace and Converter Slags, Mass %
Use of Alternative Materials in Cement Manufacturing
Prior to their use, the undesired large iron pieces were magnetically removed from the slags. Several raw feeds were prepared by mixing the slags with calcined limestone. For the two best trial burns conducted, the additions of blast furnace slag and converter slag were 49% and 8%, and 42% and 27% by mass respectively; the remainder being the calcined lime. The properties of clinkers and cements produced from the mixes were comparable to those produced commercially. The compres-sive strength obtained for the cement exceeded the minimum required by the ASTM C 150 specifi-cations. Their compositions suggest that the slags can be used in large portions provided the formulation of kiln feed meets the desired clinkering parameters.
Blast Furnace Barium-Manganese Slag Mixtures
Valkova and Dogandzhieva (1980) also used blast furnace slags (both crystallized and vitreous) as raw mix for producing cement clinker. These slags, however, contained higher contents of barium and manganese oxides. It was demonstrated that up to 20% of these slags could be used in the raw mix. The slags enhanced the reactivity of the raw mix and improved clinker formation. The pres-ence of barium and manganese oxides modified the alite crystals and produced increased compres-sive strength.
Phosphorus Furnace Slag
Phosphorus furnace slag is a by-product of phosphate processing. Production of one ton of phos-phorus generates about seven tons of slag. The slag is typically rich in calcium silicates, with phosphorus (2.5%) and fluoride (1.5%) as minor constituents. Singh and Bhattacharjee (1996) made use of 2.5% granulated phosphorus furnace slag (GPFS) in the raw mix of vertical shaft kiln in cement manufacture. The use of GPFS improved the quality of the clinker and produced cement with better strengths than the control. To the extent that such slags can replace limestone, they have the potential to save calcination energy.
Oil Shale Residues
A Russian cement plant used lime-rich oil shale residues in the manufacture of cement (Freiman and Kougija, 1997). The residues contained between 34.5% to 48.9% CaO and 28% to 38.2% SiO2. The calcareous component used was waste limestone that contained between 2% to 6% organic substances. The kiln feed was prepared in a mixing chamber of the first stage of the cyclone pre-heater. Oil shale was added in the calciner or the riser duct at about 8% of the raw mix. The resid-ual fuel in the shale gave substantial energy savings and the clinker homogeneity, as examined by microscopy, was adequate. This study was a sequel to earlier work in Germany at Rohrbach’s Dotternhausen plant (Rohrbach, 1969; Rechmeier, 1970), in which 8% of the kiln feed was oil shale added directly to the calcining process, and oil shale ash was interground with clinker to create blended cements without the need for gypsum addition.
MATERIALS REPLACING SILICEOUS COMPONENTS
This section focuses on wastes used for replacing siliceous components (containing silica) in cement raw feed.
Foundry Sand
Foundry sand is a waste from metal processing foundries. It is used in making molds for casting molten metal and alloys. Foundry sand is primarily rich in silica, with a small percentage of clay and molasses. Foundry sand has been used to replace silica in cement kiln raw feed; however, its use depends upon the composition of the foundry sand and the target kiln feed mix-design
(Bhatty, 2002). A typical clay-bonded foundry sand composition is shown in Table 2.4.10.
Table 2.4.10. Composition of Foundry Sand (Clay-bonded), Mass %
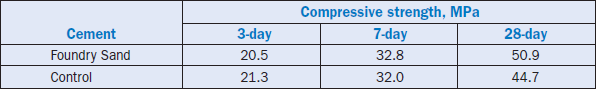
Foundry sand was used to totally replace silica in the cement raw feed. The clinker produced has a phase distribution typical of Type I/II clinkers as shown in Figure 2.4.4. The addition of foundry sand made the clinker somewhat harder to grind. The low grindability can be attributed to the presence of large alite and belite crystals formed due to coarse quartz in the foundry sand. The late strength of cement was better than that of control clinker. The 3-, 7-, and 28-day compressive strength of cement produced from the foundry sand versus that of a control cement is shown in Table 2.4.11.

Figure 2.4.4. Clinker made from raw feed containing foundry sand.
Table 2.4.11. Compressive Strength of Cement from Foundry Sand Clinker
Sand Washings
Sand washings are traditionally rich in silica, ranging up to 75% silica by mass. The other compo-nents are largely clay. Owing to their compatible composition, these washings could find a reason-able use as an ingredient in the raw mix for cement manufacture.
Rice Husk Ash
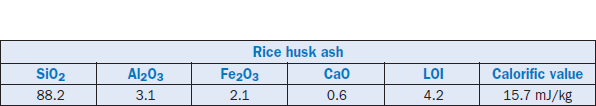
Rice husk ash (RHA) is produced by incinerating rice paddies. The ash is rich in silica; it also contains some unburned carbon. The ash has been successfully used as a kiln feed supplement to partially substitute for high-ash coal in the dry kiln process with a suspension preheater (Ghosh and others, 1992). The composition of the ash used is shown in Table 2.4.12. At another cement plant, rice husk ash was used as an alternative to sandstone in a raw mix prepared with high-grade limestone. The use of RHA improved the kiln feed burnability and eased the strength and expan-sion problems in cement performance. In both cases the ash also contributed to fuel savings because of the unburned carbon.
Table 2.4.12. Chemical Composition of Rice Husk Ash Used (Mass %) and Heat Value
Substitution of coal ash by 10% RHA contributed 5% to the fuel economy with no adverse effects to the clinker and cement properties. Additionally, the compressive strength of the resulting cement increased. A 20% substitution of RHA contributed to about 10% economy in fuel cost per ton of clinker. However, the clinker quality was adversely affected, at least in part because of a severe decrease in the amount of alite, and required blending with high quality clinker to produce cement with acceptable properties. The mix may not have been optimally proportioned, in view of the reduced alite content. Select properties of clinker and cement made with RHA substitution are given in Tables 2.4.13.
Table 2.4.13. Properties of Clinkers and Cements Made With RHA Substitution in Raw Mix, Mass %
Singh and others (1997) also used RHA as a raw feed component in a vertical shaft kiln, and produced clinkers and cements of acceptable quality. An addition of 3% RHA enhanced the kiln feed burnability, improved the production rate by 10%, and gave noticeable fuel savings. Additionally, the presence of minor constituents like phosphate and sulfates in the RHA could increase the rate of clinkering by their mineralizing action, whereas the low magnesia content could help cement plants using high-magnesia limestone to mitigate problems related to sound-ness. Ajiwe and others (2000) used RHA to produce white cement. In one study, 24.5% RHA was used with other raw materials, and the cement produced was evaluated for chemical as well as physical properties. The data confirmed that the cement produced from the RHA combination was similar to the commercially produced cement.
Ore Rejects and Mineral Tailings
Tailings from ores and mineral processing plants are also rich in silica. One study has reported the use of copper-nickel and taconite (iron ore) tailings as replacements for silica in cement raw mix (Bhatty and others, 1985). The copper-nickel tailings were beneficiated prior to their addition to the raw mix. Their composition is shown in Table 2.4.14.
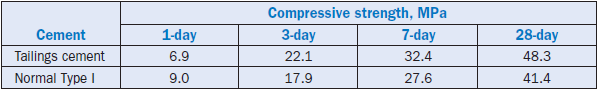
The clinker produced from the raw mix was a Type I/II. It showed normal distribution of the major phases. The strength properties of the cement were better than those of a normal commer-cial Type I cement as shown in Table 2.4.15.
Table 2.4.14. Composition of Mineral Tailings, Mass %
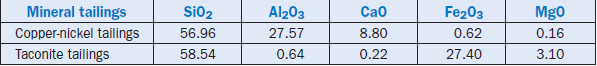
Table 2.4.15. Strength of Tailing Cement and Normal Type I Portland Cement
MATERIALS REPLACING ARGILLACEOUS COMPONENTS
This section discusses wastes and by-products that replace argillaceous components (silica and alumina) in the raw mix used in cement manufacturing. Materials discussed in this category include catalyst fines, fly ash, bottom ash, pond ash, red mud, basalt rock, and others.
Catalyst Fines
Catalyst fines are traditionally rich in silica and alumina. A typical composition of catalyst fines had 50% SiO2 and 40% Al2O3 (Bhatty, 2001). A 10% addition of these fines was made to replace a large portion of silica and alumina (nearly 25%) in a raw mix to produce cement clinker. The analyses indicate that mostly Type I/II clinker was produced from these mixes. The clinker showed phase distribution similar to that of the control (made without the catalytic fines). Bogue analysis of clinker made with catalyst fines compared to the control is shown in Table 2.4.16. The addition of catalyst fines in the raw mix made clinker harder to grind, probably due to the formation of somewhat larger alite crystals.

Figure 2.4.5. Clinker made from raw feed containing catalytic fine.
Table 2.4.16. Bogue Analyses of Clinker Made With Catalyst Fines vs. Control
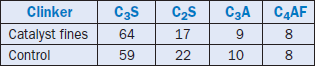
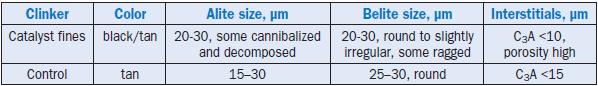
Table 2.4.17. Microscopic Observations of Clinkers Containing Catalyst Fines vs. Control
The compressive strength of cement pastes made with the catalyst fines clinker relative to the control is shown in Table 2.4.18.
Table 2.4.18. Strength of Cement from Catalyst Fines Clinker vs. Control
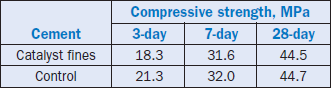
There appears to be no change in the initial setting time of cement paste made with the catalyst fines clinker. The early strength is somewhat low, but the 7- and 28-day strengths are essentially equal to that of the control.
Fly Ash
Fly ash is produced from coal-fired power plants. Presently well over 60 million tons fly ash are produced annually in the U.S.A. Only a portion of the ash is reused into marketable products, while the rest (over 40 million tons) is discarded. Figure 2.4.6 shows piles of preconditioned (with lime) waste fly ash (prior to landfilling) generated at one of the Midwest power plants in the United States.
Being rich in silica and alumina, fly ash is regarded as a compatible component of cement raw feed largely replacing clay or shale in the original feed. Typically, the composition of fly ash is similar to that of clay and shale except that it generally contains more alumina. However, in some cases, a high content of iron oxide in fly ashes can also be the sole source of iron required by the raw mix.

Figure 2.4.6. Waste fly ash generated at a coal-fired power plant.
Several cement plants in the U.S. have made use of fly ash as a component of raw feed. Plant types, use rate, and mode of fly ash addition are given Table 2.4.19.
Table 2.4.19. Plant Types, Fly Ash Addition, and Mode of Use in U.S. Cement Plants

In all these plants, the use of fly ash in raw kiln feed produced normal clinkers showing satisfactory amounts of C3S, C2S, C3A, C4AF, and liquid phases in the final clinker.
At one of the largest cement plants in North America, the use of shale was completely eliminated by replacing with fly ash and iron ore tailings (Everett, 1995). The practice successfully reduced the generation of cement kiln dust (CKD). Prior to the switch over, the shale used at the plant was oil-bearing and contained about 5% sulfur that caused the plant to generate a large amount of CKD. By using the right proportions of fly ash (as a source of alumina) and iron ore (as a source of iron oxide) shale was replaced and CKD was reduced by at lest 50% while producing a low-alkali cement. This was in addition to a significant reduction in both SO2 and hydrocarbon emissions from the plant stacks.
Ålborg Portland cement plant in Rørdal, Denmark, introduced 10% fly ash directly into the burn-ing zone (Miller, 1980). The clinker was of good quality, showing normal phase distribution. Later in 1988, an improvement in the feeding system of fly ash in the raw kiln material and installation of a modern precalciner plant by Ålborg, allowed an increase in the proportion of fly ash in the clinker to 24% without any detrimental effects to the quality of the clinker or to the environment (Borgholm and Nielsen, 1989). Reportedly, 2.3 million tons of fly ash have been processed,of which more than 50% were consumed as a raw feed component in cement manufacture
(Borgholm, 1992).
Two wet process plants in Belgium also used fly ash. Favorable change in slurry moisture, decreased dust loss, and reduced energy consumption were realized. Although the clinker showed an increased homogeneity of the major phases, it appeared overburned and hard to grind, causing grinding energy to increase. It is possible that the overburning was due in part to a failure to adequately compensate for the fuel value in the fly ash.
A Japanese cement plant used silica-rich fly ashes as a partial substitute for clay in the raw kiln feed (Mori, 1982). A treatment plant at the Takehara power station was installed to control the fly ash composition and create a sustainable supply to produce cement clinker. The chemical composi-tions of the two fly ashes (A and B) and the clay used in the raw mix are given in Table 2.4.20. The fly ash replaced 20% to 40% of the clay depending on the compositional compatibility.
Table 2.4.20. Composition of Fly Ashes and Clay, Mass %
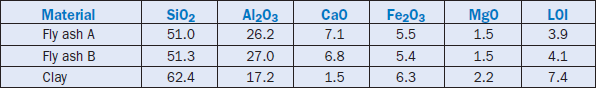
Harada (1991) also reported a case study of recovery and utilization of fly ash and its major use in clinker manufacture. Fly ash replaced clay in the cement raw mix. No problem was encountered with the granularity or the presence of limited unburned carbon in the fly ash.
Mullick and Ahluwalia (1992) reported the use of fly ash in Indian cement manufacturing. The composition of the fly ashes used was close to that of clay,which was replaced fully or partially in the cement raw mix. Gore and others (1992) replaced 6% clay with fly ash in a wet process raw feed. The modification in the raw feed brought about by fly ash substitution gave several material, operational, and energy benefits. Lime combinability of the raw mix with and without the fly ash addition is shown in Figure 2.4.7.
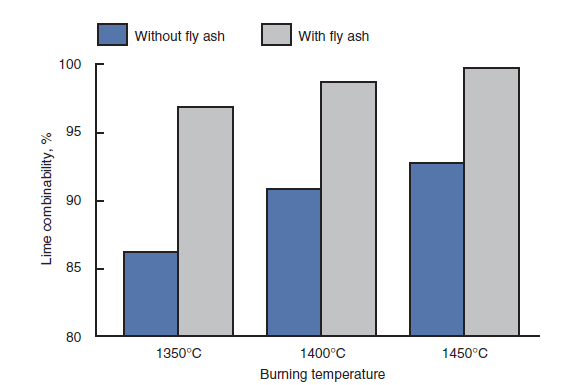
Figure 2.4.7. Lime combinability of the raw mix with and without the fly ash addition.
Kelly (1994) reported that control of the lime saturation factor, silica ratio, and alumina ratio for a target clinker was superior when raw mixes contained fly ash as compared to an equivalent amount of shale. The demonstration was done by replacing shale with 3.2% fly ash. The addition of fly ash also enhanced the C3A level of clinker, with equivalent C3S and C2S contents, as shown in Table 2.4.21.
Table 2.4.21. Raw Mix Parameters and Clinker Phases with and without Fly Ash Additions
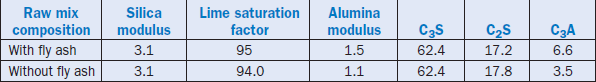
The early-age strength gain of cement prepared from these clinkers was superior to that of control. In addition, an increased C3A content allowed a larger scope for optimizing the early setting and strength properties of cement because gypsum could be added to the clinker over a wider range of concentrations during the finish grinding.
In general, the following processing, operational, and product benefits have been observed from the use of fly ash as a component in cement raw kiln feed:
• Improved burnability, resulting in lower burning temperatures and a savings in fuel
• Increased clinker production
• No apparent deterioration in clinker quality
• Reduced SO2,hydrocarbon, and NOx emissions (in wet process) for some plants and fly ashes
• Reduced generation of cement kiln dust
Since fly ash is a free-flowing particulate material which is readily mixed and blended, additional benefits have also been realized as follows:
• Reduced material processing cost
• Reduced dusting due to reduced comminution of raw materials
However, some noteworthy adverse effects of using fly ash have also been pointed out. Some fly ashes cause reduced plasticity of the feed slurry in the wet process, while in a dry process plant, the fly ash resulted in an increased volatilization of alkalis that can cause blockages in preheaters.
High-Carbon Fly Ash
With the installation of low-NOx burners and other emission-reducing measures at coal-fired power plants, large amounts of high-carbon fly ashes are expected to be generated. Although these ashes will pose difficulties for disposal, they will be capable of offering two major benefits if used in the cement manufacturing. First, the fly ash can partially replace raw materials such as shale and clay (as discussed above), and secondly, the carbon content of the fly ash can provide a fuel supple-ment during the clinker-firing process.
Bhatty and others (2000 and 2001) demonstrated the use of a fly ash that contained approximately 20% unburned carbon at a suspension preheater plant. The composition of the fly ash used in the trial run is shown in Table 2.4.22.
Table 2.4.22. Oxide Composition of the High-Carbon Fly Ash, Mass %

*Composite contains 3.53% Na2O equivalent alkalis, and has moisture content of 0.33%.
The high-carbon fly ash was blended with the raw materials (crushed limestone and a small amount of shale) and ground into raw feed. The composition of the raw feed was targeted to the normal raw feed. The fly ash composition and that of other raw materials (limestone and shale) limited the fly ash substitution to 6%. The total heat content (calorific value) of the ash was esti-mated to be greater than 740 kJ/kg, which translated to an anticipated energy contribution from the fly ash of 67 kJ/kg of clinker.
During the demonstrations, the operation ran in a more efficient, stable, and predictable manner. Consequently, the cement plant incurred a fuel savings of nearly 4%, and the clinker production increased by almost 10%.
Characteristics of clinker produced during the demonstration as determined by XRF, XRD, and free-lime tests are shown in Table 2.4.23. The XRF analysis indicates lower sulfate and alkalies in the fly ash clinker than in the control. Figure 2.4.8 shows the major phase distribution in the clinker.
Table 2.4.23. Select Oxide Composition of Clinkers, Mass %
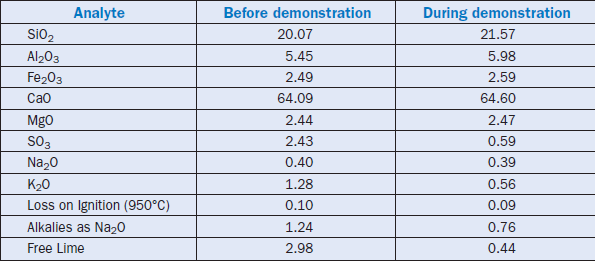

Figure 2.4.8. Clinker produced from raw feed containing high-carbon fly ash, showing typi-cal major clinker phases.
Test results and ASTM C 150 standard physical requirements for cements presented in
Table 2.4.24 indicate that the demonstration cement is comparable to the commercially produced cements.
A similar study for Electric Power Research Institute (EPRI) was carried out by Bhatty and others (1998) on several high-carbon fly ashes (carbon ranging from 6% to 16%) that produced similar results. The ashes were used as a component for raw feed collected from different cement plants across the U.S. Clinkers and cement were produced from these mixes and tested. The data indicated that clinkers had normal formation and distribution of the major clinker phases, and the cement had strength and setting properties comparable to that produced at the actual cement plants.
A cement plant in Canada has also used up to 10% high-carbon fly ash as a raw kiln feed compo-nent. The primary motivation for the plant is fuel savings.
Table 2.4.24. ASTM C 150 Data for Cements Produced Before and During the Demonstration
Bottom Ash
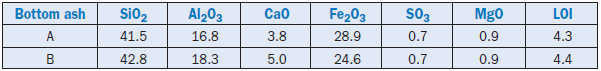
Bottom ash is also a by-product from coal-fired power plants. Typically, a bottom ash is composed of partially fused coarse-sized particles. The ash is rich in silica, iron, and alumina and is suitable as a replacement for shale and clay, and iron (as corrective material) in cement raw feed. Compositions of select bottom ashes are shown in Table 2.4.25.
Table 2.4.25. Composition of Bottom Ashes, Mass %
In the long dry and preheater cement manufacturing processes bottom ash is generally blended with the raw mix prior to grinding. In the wet process, however, the bottom ash and clay are combined during the clay-wash process and added to the raw feed. Bottom ash is typically added as 2% to 5% of the raw feed, and produces clinkers showing satisfactory distribution of C3S, C2S, C3A, C4AF, and liquid phases; cements also display acceptable chemical and physical properties.
Ponded Ash
Pond ash is generally a mixture of fly ash and bottom ash from coal-fired power plants. The mate-rial is generally sluiced and stored in ponds. Millions of tons of ash have been accumulated in the ponds over the past several years; Figure 2.4.9 shows one of the ponds holding ash generated from a power plant in the United States.
Singh and others (1995) have made use of ponded ash as a reactive component in the raw mix in a vertical shaft kiln. Use of 4% pond ash gave optimum performance during the kiln operation. Composition of the pond ash used is shown in Table 2.4.26. The ash also contained a high carbon content. The raw feed mixture containing fly ash was nodulized prior to firing.

Figure 2.4.9. Ponded combustion wastes from a coal fired power plant.
Table 2.4.26. Composition of Pond Ash Used in Cement Operation, Mass %
![]()
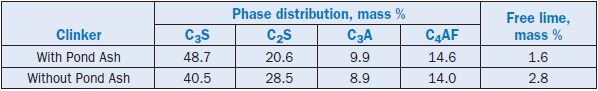
The clinker produced was of acceptable quality and was easier to grind, resulting in a savings in grinding energy. Since in a shaft kiln operation the fuel is incorporated into the raw feed, this prac-tice was well suited for high-carbon pond ashes. The use of ash also realized an improvement in clinker quality, energy conservation, and cement production for the plant. Composition of the clinker produced with and without pond ash are shown in Table 2.4.27.
Table 2.4.27. Composition of Clinkers from Raw Mixes With and Without Pond Ash
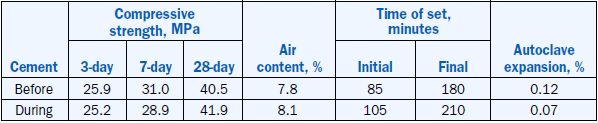
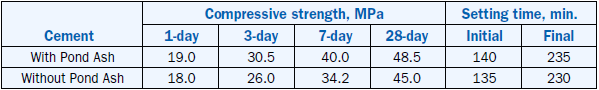
As expected, the strength properties of cement made with pond ash clinkers were better than the control clinker because of higher C3S content; setting properties were comparable. Strength and setting data is shown in Table 2.4.28.
Table 2.4.28. Strength and Setting Data of Cements from Raw Mix With and Without Pond Ash
Coal Processing Wastes
Similar to high-carbon fly ash, coal processing plant wastes (also know as coal prep wastes) are being examined for use as a raw material in cement. Currently the authors of this chapter are investigating the use of these materials as a partial raw material replacement and partial fuel substi-tute. Because they are unburned and somewhat moist, these materials may affect emissions, limit-ing their use to particular types of cement manu-facturing operations. Several million tons of coal are discarded (see Figure 2.4.10 for a coal waste disposal area). Reutilization of coal prep waste in cement manufacturing can result in high volume use with significant material and environmental benefits.

Figure 2.4.10. Landfill of coal prep-waste wastes are generated every year, most of which next to a coal mine.
Red Mud
Red mud is a waste generated when alumina is recovered from bauxite during the Bayer process. Red mud is especially rich in Fe2O3 and A12O3 and also contains SiO2.Being rich in iron, it appears red in color, hence the name. Satisfactory use of red mud as an iron source in cement raw feed has been reported by Laxmi and Parashar (1985). Oxide compositions of two red muds used at different cement facilities are shown in Table 2.4.29. The amounts of red mud used in cement raw mix are also shown in the far right column of the table.
Table 2.4.29. Oxide Composition of Red Muds Used in Different Case Studies, Mass %
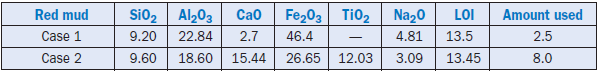
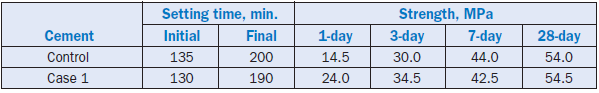
Case 1 used 2.5% red mud as replacement for iron ore in the raw mix. Cement produced from the raw mix showed acceptable setting behavior and a significant improvement in the early strengths The later strengths were also comparable to cement produced without red mud substitution (see Table 2.4.30). The red mud substitution also helped address the problem of sulfur build-up in the kiln system of the cement plant.
Table 2.4.30. Setting and Strength of Cement Made with Red Mud in Case 1
Case 2 used 8% red mud in the raw mix and produced clinker of good quality. The clinker showed well-developed major clinker phases of alite and belite. It may be noted that excessive titanium content can have an adverse effect on the formation of the melt phase, and can restrict the addition of red mud in the raw mix.
Basalt Rock
Basalt rocks are volcanic in nature, and can be reactive to promote lime combinability in cement kiln feed. Basalt is rich in silica and alumina, with other significant components being lime, iron, and magnesium. The bulk of silica in basalt is in the form of silicates; iron is present as magnetite and ilmenite; and almost all magnesia is in the form of magnesium ferrosilicate. A range of typical basalt compositions is shown in Table 2.4.31.
Table 2.4.31. Chemical Compositions of Several Basalt Rocks, Mass %
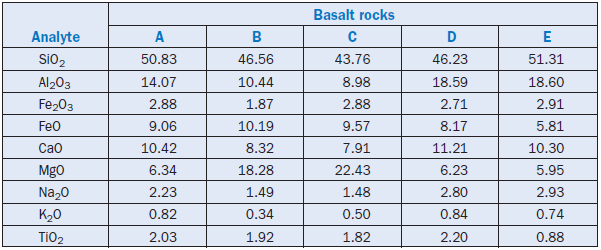
Promising test results on the use of basalt as a corrective material in the raw feed for portland cement manufacture have been reported by Khadilkar and others (1989). Improvement in the raw mix reactivity was studied by firing raw feed containing basalt. The raw feed was prepared with 2%basalt as the corrective material by replacing bauxite and iron ore in the feed. The material was pelletized and fired from 800°C to 1450°C for 20 minutes. The fired pellets were tested for free lime and also for any expansion or shrinkage; their mineralogical characteristics were determined by the XRD analyses.
Raw mix containing basalt underwent more expansion than the mix without basalt up to 1100°C, suggesting higher rates of solid-solid reactions to form calcium silicates and aluminates. Higher shrinkage of basalt raw mix above 1300°C suggests higher liquid formation and C3S formation. Significant reduction in free lime for the basalt-containing mix also indicates improved lime combinability with basalt. The XRD analysis confirm the formation of C3S at around 1300°C for basalt-containing raw feed as compared to the raw mix without basalt which showed the C3S peak at a much higher temperature.
The microstructure of clinker produced from basalt raw feed exhibited well-developed C3S and C2S crystals. Stabilized polymorphs of belite were observed with higher hydraulic activity. Consequently the cement produced from basalt clinker gave higher compressive strength.
OTHER MATERIALS
As mentioned above, other materials that do not replace the calcareous, siliceous, or argillaceous materials can also be used in cement manufacture.
Wastes with Fluxing and Mineralizing Potentials
Wastes from the fluoride and fertilizer industries contain components with mineralizing and flux-ing capability. The principal component in these wastes is calcium fluoride or its compounds which have been used advantageously in the cement industry. Use of waste from zinc, molybde-num, barium, and phosphogypsum industries has also been reported. The wastes have been added to the raw mix to promote reactivity at reduced clinkering temperature. When used as a mineral-izer, zinc waste improved the burnability of cement raw mix. Figure 2.4.11 shows free lime as a function of ZnO in the waste added to the raw mix. Zinc-containing waste was also an effective mineralizer for the raw mix in the vertical shaft kiln process; it activated the formation of alite in clinker and improved early strength of the cement (Jinyu and others, 1989).
Molybdenum-iron ore tailings as studied by Zhou (1992) also imparted mineralizing effect on the clinkering of cement raw feed. It promoted clinker formation by reducing the melt viscosity and helped accelerate C3S formation. When used in the plant, the tailings produced clinker and cement with excellent properties. Free lime in clinker decreased, and the early age and ultimate strength of cement increased significantly. It markedly increased the kiln output by greater than 13%, and improved fuel saving by over 5%.
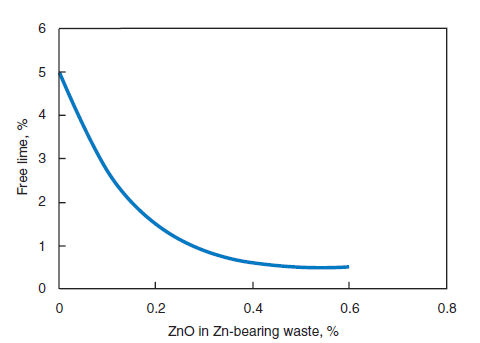
Figure 2.4.11. Free lime as a function of ZnO in the waste added to the raw mix.
Flue Gas Desulfurization Sludge
Flue gas desulfurization (FGD) sludge is collected from the scrubbers of the power plants that use high sulfur coal. Lime is made to react with the SO2 released from the coal to form calcium sulfite or calcium sulfate sludge that is collected by the scrubbers. FGD sludge is a fine material collected from the filtration units; Figure 2.4.12 shows collection of sludge form filtration drums. Typical FGD sludge is mainly calcium sulfite (CaSO3·1⁄2H2O). Some power plants have additional equipment to oxidize the FGD sludge so that the calcium sulfite is converted to the more marketable calcium sulfate (gypsum).

Figure 2.4.12. Flue gas desulfurization (FGD) sludge collected from filtration drums at a power plant.
Since, the oxidized FGD sludge is 99% gypsum, it can conveniently be used as a gypsum substi-tute in the finish milling of clinker in a cement plant to produce cement. The sludge is also a finely divided material that provides energy relief during finish milling. It also saves energy other-wise required to crush gypsum prior to the finish milling. Handling issues, however, need to be addressed before successful implementation at a plant. The sludge is often available in moist condition. It’s adequate drying frequently resulting in excessive flowability can be a problem, in addition to concerns relative to outside storage.
No adverse effects on the setting or the strength properties of cement have been reported from cements made with oxidized FGD sludge. These cements meet the ASTM C150 standard specifica-tions for both physical and chemical requirements.
CONCLUDING REMARKS
Owing to the compositional compatibility of several wastes and by-products with cement raw materials, their potential for reutilization in cement manufacture is substantial. However, their use largely depends on their chemical composition and compatibility with the materials they replace in the cement raw mix.
The cement industry has been making use of numerous wastes and by-products as raw feed components while tracking the economic, operational, and environmental ramifications to cement
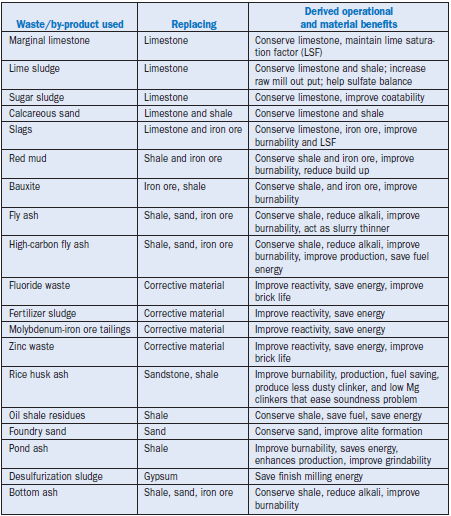
manufacturing. It is extremely critical, as mentioned above, that the composition of these materi-als be compatible with those of the raw materials they replace in the cement raw mix. Based on this criterion, the materials identified as raw material components in cement manufacture include marginal limestone, lime-bearing carbonates, sludges, metallurgical slags, fly ash, bottom ash, desulfurization sludge from coal-fired power plants, mine and mineral wastes, catalyst fines, and foundry sand. A summary of commonly used wastes and by-products in the cement industry is given Table 2.4.32, while the derived operational and material benefits realized during cement manufacturing are given in Table 2.4.33.
Table 2.4.32. Waste and By-Products Used in Cement Manufacture

Table 2.4.33. Material and Operational Benefits of Using Wastes and By-Products in Cement Manufacture
REFERENCES
Ahluwalia, S. C., and Page, C. H., “Effect of Low Grade Fuels, Combustibles, and Non-Traditional Raw Materials,” 9th International Congress on Chemistry of Cement, New Delhi, India, Vol. II, 1992, pages 83-122.
Ahluwalia, S. C.; Laxmi, S.; and Chopra, S. K., “High Magnesia and Marginal Grade Limestone Prospects, Problems and Technology of Utilization,” All India Seminar on Cement Manufacture, Vol. I, 1981, pages 47-79.
Ajiwe, V. I. E.; Okeke, C. A.; and Akigwe, “A Preliminary Study of Manufacture of Cement from Rice Hush Ash,” Biresource Technology, Vol. 73m, 2000, pages 37-39.
Bhatty, J. I.; Marrijnissen, J.; and Reid, K. J., “Portland Cement Production Using Mineral Wastes,” Cement and Concrete Research, Elmsford, New York, U.S.A., Vol. 15, 1985, pages 501-510.
Bhatty, J. I., “Role of Trace Element on Cement Manufacture and Use – A Laboratory Study,” Portland Cement Association, Skokie, Illinois, 2002, 40 pages.
Bhatty, J. I.; Campell, D. H.; Detwiler, R. J.; Eddinngs, E. G.; Gajda, J.; Miller, F. M.; Mishulovich, A.; and Taylor, P., “Use of High Carbon Fly Ash as Component of Raw Mix for Cement Manufacture,” Final Report No. WO 6661-01, Electric Power Research Institute (EPRI), Palo Alto, California,May1998.
Bhatty, J. I.; Gajda, J.; and Miller, F. M., “Use of High Carbon Fly Ash in Cement Manufacture,” Cement Americas, May/June, 2001, pages 32-34.
Bhatty, J. I.; Gajda, J.; and Miller, F. M., “Use of High Carbon Illinois Fly Ash in Cement Manufacturing,” Final Report No. 99-1/2.1A-1, Illinois Clean Coal Institute (ICCI), Carterville, Illinois, 2000.
Borgholm, H. E., “Reducing Environmental Pollution by Using Fly Ash as a Raw Material Component,” Zement-Kalk-Gips, Bauverlg GMBH/Maclean Hunter, Wiesbaden, Germany, Vol. 45:4., April 1992, pages 163-170.
Borgholm, Hans E., and Nielsen, Peter B., “Ålborg Portland Converts to the Semi-Dry Process,” IEEE Transactions on Industry Applications, Vol. 25 No. 3, May-June 1989, pages 486-494.
Du Toit, Pieter, “The Use of Tswana-Only Limestone for Clinker Production at the Blue Circle Cement Lichtenburg Plant in South Africa,” Proceedings of 18th International Conference on Cement Microscopy, Houston, Texas, U.S.A., Vol. 18, 1996, pages 134-148.
Everett, D., “Raw Materials are the Key to CKD Reduction,” Rock Products, Cement Edition, July 1995, pages 16-21
.
Freiman, L. S., and Kougija, M. W., “Residues from Oil Shale Processing Used as Cement Material,” Zement-Kalk-Gips, Bauverlg GMBH/Maclean Hunter, Wiesbaden, Germany, Vol. 50, 1997, pages 44-49.
Fuller, Fuller Kiln Dust Recovery System, Bethlehem, Pennsylvania, U.S.A., 1992.
Ghosh, S. P.; Mohan, K.; and Gandhi, R. K., “Effect of Using Rice Husk and Its Ash as Fuel and Raw Material Component Respectively in Cement Manufacture,” 9th International Congress on Chemistry of Cement, New Delhi, India, Vol. II, 1992, pages 224-231.
Gore, V. K.; Khadilkar, S. A.; Lele, P. G.; Page, C. H.; and Chatterjee, A. K., “Conservation of Raw Material Through Use of Industrial Wastes in Cement Manufacture,” 9th International Congress on Chemistry of Cement, New Delhi, Vol. II-Theme I, 1992, pages 238-244.
Harada, M., “Recovery and Utilization of Coal Ash,” Shigen To Sozai, Japan, Vol. 107:2, February 1991, pages 85-91.
Jinyu, Zhong; Yueming, Fan; Hui, Lu; and Jiazhi, Vang, “Study on Using Zn Containing Waste as Mineraliser,” Beijing, Vol.2, 1989, pages 346-352.
Kelly, R. J., “Coal Ash – A Useful Raw Material for Portland Cement Manufacture,” Ash – A Valuable Resource, Second International Symposium, Volume 2, Feb. 1994, South African Coal Ash Association Publication, Cresta, South Africa, 1994, pages 373-380.
Khadilkar, S. A.; Ghosh, D.; and Page, C. H., “Investigation on the Use of Basalt as a Corrective in Portland Cement Manufacture – A Case Study,” 2nd NCB International Seminar, Vol. II, 1989, pages Vl-l2.
Laxmi, S., and Parashar, A. K., “Potentials of Utilisation of Wastes from Fertilizer and Aluminum Industries for Building Materials: An Overview,” National Seminar on Role of Building Materials Industries in Conversion of Waste into Wealth, Vol. II, March 1985, pages 101-111.
Miller, F. M., “Effect on Clinker Quality of the Fly Ash Insufflation, Kiln No. 86,” Report No. 3: Evaluation of New Fly Ash Feeder, Low Slurry Moisture Level, and Reduced Kiln Feed Rate. Evaluation of Two Belgian Clinkers Produced with High-Ash Fuel, F.L. Smidth, Røderal, Denmark, April 1980.
Monshi, A., and Asgarani, M. K., “Producing Portland Cement from Iron and Steel Slags and Limestone,” Cement and Concrete Research, Elmsford, New York, U.S.A., Vol. 29, No. 9, 1999, pages 1373-1377.
Mori,T., “Features of Coal Storage and Coal Ash Treatment Systems for Large Scale Coal-Fired Power Generation at EPDC’s Takehara Thermal Power Station, No. 3 Extension Unit,” Sixth International Conference on Coal Research, National Coal Board, London, Paper F6, 1982,
pages 705-725.
Mullick, A. K, and Ahluwalia, S. C., “Utilization of Wastes in Indian Cement Industry,” Cement Industry Solutions to Waste Management, First International Symposium, Calgary, Alberta, Canada, October 1992, pages 489-512.
NCB “Evaluation of Red Mud for its Utilisation in the Manufacture of OPC,” Sponsored Project Report, March 1989.
NCB, “Suitability of Carbide Sludge for the Manufacture of Cement,” Sponsored Project Report, March 1987.
NCB, “Technical Suitability of Using Calcium Carbonate Sludge as a Part Substitute to Limestone for the Manufacture of Ordinary Portland Cement,” Sponsored Project Report, October 1990.
Puertas, F. M.; Vareta, Blanco T.; Palomo, A.; and Vazquez, P., “Reactivity and Burnability of Raw Mixes Made With Crystalised Blast Furnace Slags,” Zement-Kalk-Gips, Bauverlg GMBH/Maclean Hunter, Wiesbaden, Germany, Part I, 8, 1988, pages 398-402.
Purohit, M. A.; Rasal, S. M.; and Shaha, R. D., “Kiln Feed Filtration for Semi Dry Conversion,” First NCR International Seminar on Cement and Building Materials, New Delhi, India, Vol.2 III, 1987, pages 59-74.
Raina, K.; Nedumaran, B.; and Mehrotra, S. N., “Utilization of Kiln Dust for Clinker Production Using CRI-Modern Vertical kiln Technology,” 10th International Congress on Chemistry of Cement, Gothenburg, Sweden, Vol. 1, Paper 1i002, June 1997, 4 pages.
Rechmeier, H., “The five stage preheater kiln for burning of clinker from linestone and oil shale,” Zement-Kalk-Gips 23, 1970, pages 249-253.
Rohrbach, R., “Production of Oil Shale Cement and Recovery of Electrical Energy from Oil Shale Using the Rohrbach-Lurgi Process,” Zement-Kalk-Gips, 22, 1969, pages 293-296.
Shrinkhande, B. K.; Tiwari, T. N.; and Chatterjee, A. K., “The Conversion of ACC’s Madukkarai Works to a Semi-Wet Process,” World Cement, Vol 22, No.10, October 1991, pages 18-21.
Singh, N. B., and Bhattacharjee, K. N., “Phosphorous Furnace Slag – A Potential Waste Material for the Manufacture of Cements,” Indian Journal of Engineering and Material Science, Vol. 3, February 1996, pages 41-44.
Singh, N. B.; Bhattacharjee, K. N.; and Shukla, A. K., “Rational Utilization of Rice Husk Ash in Mini-Cement Plants,” Zement-Kalk-Gips, Bauverlg GMBH/Maclean Hunter, Wiesbaden, Germany, Vol. 86, No. 10, October 1997, pages 594-600.
Singh, N. B.; Bhattacharjee, K. N.; Shukla, A. K., “Pond Ash – A Potential Reactive Raw Material in the Black Meal Process of Cement Manufacture by Vertical Shaft Kiln (VSK) Technology,” Cement and Concrete Research, Elmsford, New York, U.S.A., Vol. 25, No. 3, April 1995, pages 459-464.
Valkova, I. S., and Dogandzhieva, R. G., “Producing Portland Cement Clinker Using Blast Furnace Barric- Manganese Slag,” 7th International Congress on Chemistry of Cement, Paris, Vol. II, 1980, pages I:79-83.
Young, G. L., and Morrison, G. L., “Passamaquoddy Technology Recovery Scrubber at the Dragon Product Company, Inc. Cement Plant,” Rock Product, 27 International
Cement Seminar, Atlanta, Georgia, U.S.A., December 3, 1991.
Young, Rom, D., “Method and Apparatus for Using Blast Furnace Slag in Cement Clinker Production,” United States Patent 5,494,515, February 27 1996, Assignee: Texas Industries, Inc., Dallas, Texas, U.S.A.
Zhou, P. Y., “Studies on Producing Portland Cement with Tailings of Molybdenum-Iron Ore,” 9th International Congress on Chemistry of Cement, New Delhi, India, Vol. II, 1992, pages 271-277.