Contents
Emissions from cement kilns

If You Want to Download the Complete Cement Kiln Chemistry and All Complete courses and Expert Books in Cement Industry , Kindly click here NOW
Introduction
In module 3 of the course we are going to focus on the emissions from cement kilns, a subject which is attracting as much attention as energy efficiency, productivity and clinker quality is in many parts of the world..
This attention on kiln emissions has in part been invited by the trend to burn alternative fuels in cement kilns, the same emissions standards are applied to cement kilns as incinerators, despite the different operating conditions.
Concerns regarding dioxin, furan, mercury and other heavy metal emissions arise from the trend towards burning alternative fuels. The normal levels of dioxin, furan and mercury emission from cement kilns are minimal, but if alternative fuels are going to be burnt then knowledge of the mechanism of their formation and how to minimise their likelihood is required.
Most cement manufacturers will be aware of the possible global warming implications of the CO2 emissions from cement kilns. In Europe this is at the top of the agenda due to the EU Emissions Trading Scheme.
Current CO2 reduction strategies involve improved energy efficiency, replacement of fossil fuels with biomass fuels and production of blended cements to reduce the clinker content of cement. All these will reduce CO2 emissions and will enable the obligations under the Kyoto protocol to 2012 to be satisfied. These strategies are not unpopular because they also reduce costs or increase revenues for cement companies. But what about beyond Kyoto? when further reductions in CO2 emission from cement clinker manufacture will require the application of very expensive technologies?
Deforestation and the death of lakes brought about by acid rain, coupled with pollution caused by photochemical smog, have heightened awareness of the dangers of the acid gas emissions NOx and SOx. The deployment of ever more expensive technologies to reduce or mitigate against NOx and SOx emissions is demanded of the cement industry.
Increased environmental awareness by the public at large has heightened concerns with any cement factory emissions plume. Explaining this away as the condensation of water vapour may not be accepted as satisfactory, particularly if a haze is visible beyond the steam plume.
Of course dust emissions have long been associated with cement manufacture, although modern electrostatic precipitators and bag
houses are capable of reducing dust emissions to a few milligrams per Nm3 of cement kiln exhaust. In this cement kiln chemistry course we will primarily focus on the gaseous emissions from cement kilns, although there are chemistry related issues with the dust and dust handling.
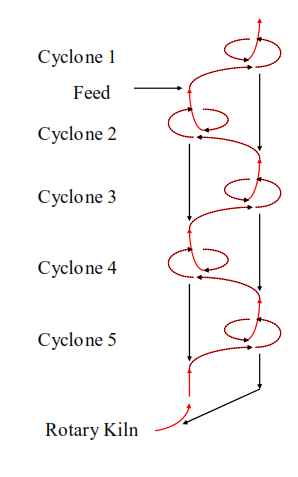
The dust collected from the kiln exhaust gases has escaped from the preheater of a modern cement kiln. It is the finest fraction of the kiln feed which is the most difficult for the preheater cyclones to collect. This physical separation means that the dust almost inevitably has a different composition to the kiln feed, usually it is higher in lime saturation as the silica concentrates in the coarser fractions of the kiln feed.
Where raw materials are dried in a drum dryer behind the kiln this may not be the case.

The handling of the dust can therefore affect the variability of the kiln feed. If added to the homogenisation silo then the affect of a layer of dust in the silo when the raw mill is stopped must be considered. Returning directly to kiln feed avoids this problem, but raises concerns of feeding two streams of material to the kiln, homogenised raw mill product and dust direct from the collection equipment.
As cement manufacturing chemists we must remember that we cannot create mass. Whatever chemical components enter the system in the raw materials fed to the raw mill and the fuels, must exit the system in the clinker or as gases in the exhausts. The composition of the kiln feed is not important as it contains raw mill product plus the dust and is the composite material in the external cycle. The inputs to the mass balance are the raw materials and the fuels, chemistry control should focus on the raw materials and raw mill product, not the composite including the dust.
We saw in session 1.7 that the alkalis and particularly chlorides can build up in an external cycle between the preheater and the dust collector. This concentration of alkalis in the dust affects the performance of electrostatic precipitators and baghouses.
The efficiency of electrostatic precipitators is dependent on the resistivity of the dust to be collected, among other things.
High alkali dust are more difficult to collect, meaning that the dust from different banks of electrostatic precipitators may have different alkali contents as the easier to precipitate low alkali dusts are collected first. On long kilns this has been used as a means to selectively extract alkalis from the process, only the dust from the final bank of the precipitator was rejected with the dust from the other banks recycled to the process. If any course participants have analyses of dust from different banks of their precipitators it would be interesting to see the results and verify if this affect is present.
The efficiency of baghouses is also affected by properties of the dust to be collected. In this case the hygroscopic characteristics of high alkali dusts is the concern and the danger of bags becoming blinded by the dust absorbing water vapour.
That’s all we will say regarding dust, we will be focusing on the gaseous emissions from cement kilns in the remainder of the course. Again I would suggest completion of Quiz 3.1 as a first step and then repeating later to gauge how much you have learned from the course.
CO2 emissions and their reduction
Cement kilns are the source of ~5% of man-made CO2 emissions in the world with CO2 being considered to be the major cause of global warming due to its trapping the sun’s energy in the atmosphere in a so-called green-house effect.
Countries in the developed world that have ratified the Kyoto protocol are committed to reducing green-house gas emissions to 87.5% of 1990 levels by 2012. As the second largest industrial CO2 emitter (after power generation) the cement industry certainly has to make its contribution to achieving this commitment.
In session 1.10 we have seen that the CO2 from calcination and the CO2 from combustion amounted to 0.857 tonnes per tonne of clinker.
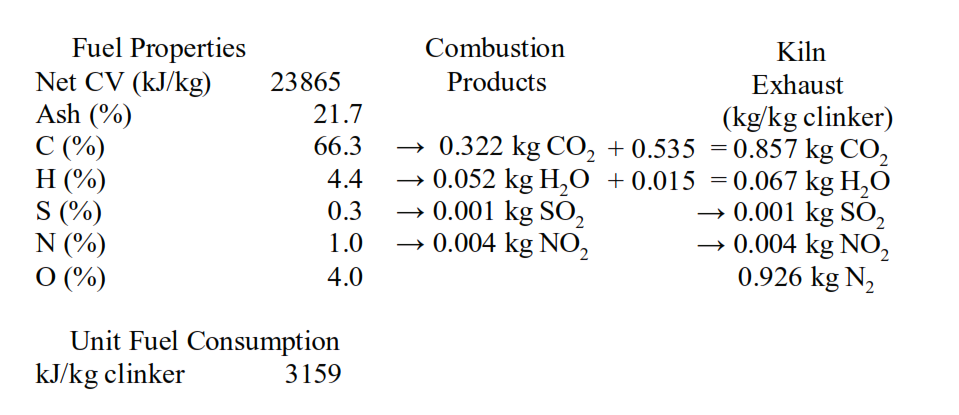
Firing the kiln with natural gas would reduce this CO2 emission by 14% to 0.74 tonnes per tonne of clinker, due to the low carbon content and higher hydrogen content.
Most cement companies have no problem in subscribing to the Kyoto target, whether they are operating in a country that has ratified the protocol or not. This level of savings can be achieved by improved energy efficiency, substituting fossil fuels with biomass fuels and production of blended cements.
These are all popular strategies with cement companies as they either reduce manufacturing costs or increase revenues. Improved energy efficiency saves costs for kiln fuel or electrical power, all cement companies can subscribe to these objectives.
Burning alternative fuels is popular, particularly if a cement company will be paid to burn the fuels. Some cement companies achieve fossil fuel replacement as high as 80%. Revenues can be significantly boosted.
Biomass fuels such as wood waste, rice husks, meat and bonemeal, or sewage sludge are treated as carbon neutral under the European Union Emissions Trading Scheme (EU ETS).
Wet process kilns with their inherently higher fuel consumption can burn more alternative fuels per tonne of clinker produced – attractive if replacement levels are high.
Production of blended cement involves replacing some of the clinker in cement with supplementary cementitious materials, limestone, pozzolans, fly ash, granulated blast furnace slag. Less clinker in cement means less CO2 emissions per tonne of cement. Granulated slag can replace over 60% of the clinker in cements for special applications. Blended cements are genuinely better than pure Portland cements in some applications, capacity and revenues of cement companies are also significantly boosted.
The real challenge for cement companies will arrive post- Kyoto. When the level of CO2 reductions required will demand the application of technologies that will significantly increase manufacturing costs.
Cement companies who must satisfy their shareholders cannot be expected to increase their costs voluntarily, some form of compulsion is likely to be required. This is the background to the EU Emissions Trading Scheme (EU ETS). Only when the cost of carbon rises significantly will commercial companies be able to justify the deployment of the technologies that will deliver the dramatic reductions in CO2 that will be required.
What might these technologies involve?
Returning to the CO2 emissions in the example from session 1.10 we can see that there is the 0.322 t/t of clinker associated with combustion of the fossil fuel. This can be reduced by 36% to 0.205 t/t by firing kilns with natural gas.
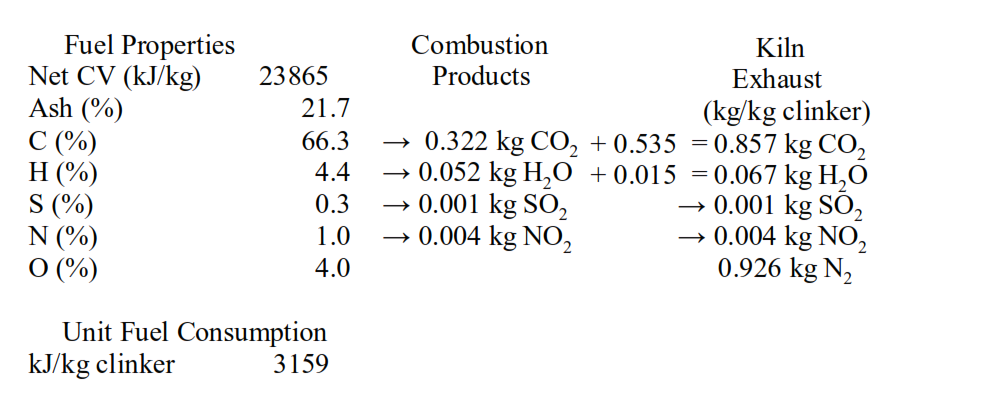
Switching to natural gas firing will not be practical for many cement companies.
The greater part of the CO2 emissions in the example from session 1.10 is the 0.535 t/t of clinker associated with calcination of CaCO3, the so-called ‘process’ CO2. It is argued that this ‘process’ CO2 is an inevitable consequence of cement manufacture. However, we have seen in session 2.10 that production of active belite cements with LSF of ~80 can reduce this by ~15% to 0.45 t/t of clinker. In combination natural gas firing and production of active belite cement would reduce CO2 emissions to 0.655 t/t clinker.
However, switching to the production of active belite cements would inevitably involve changes in the hydraulic performance of cements, in competitive markets this would represent a considerable marketing challenge. Only when the cost of CO2 emissions rises significantly will cement companies be able to take that market risk. 0.655 tonnes of CO2 per tonne of clinker remains a significant CO2 emission.
In the long term all industrial CO2 emissions are likely to be challenged. What options would then be available to cement manufacturers?
One possibility that is discussed is the combination of power generation and cement manufacture using fluidised bed kilns. Such fluidised beds could be fed with fine CaCO3 and high ash fuels proportioned together to give the required clinker composition.
Kawasaki Heavy Industries have developed a 200 tpd fluidised bed cement kiln that cuts thermal energy consumption by 10~25%, they claim this can be scaled up to 3000 tpd.
But the trend in cement kilns is to ever larger capacities to achieve economies of scale, smaller units would only be attractive if carbon costs rise. Such combined fluidised bed kilns would continue to emit CO2. The only technology for complete elimination of CO2 emissions is sequestration.
Sequestration involves the storage of CO2 in underground reservoirs by injection of CO2 into depleted oil wells, with one of the key requirements being a high concentration of CO2, but we have seen that cement kiln exhaust is less than 50% CO2 by weight…
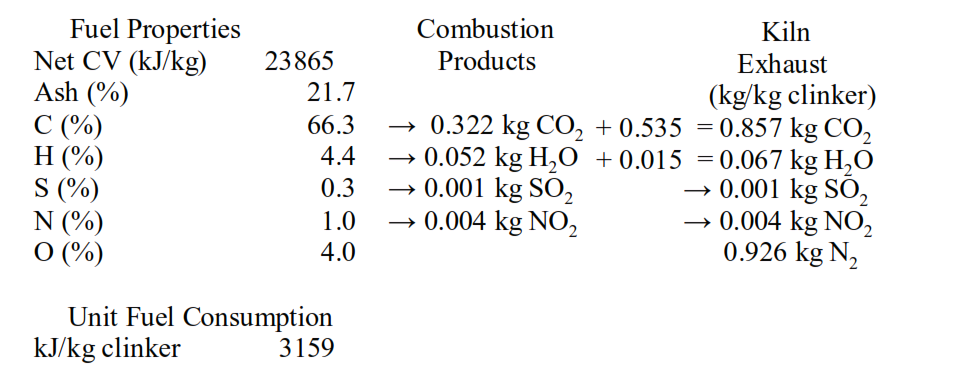
and only ~35% by volume, with excess air this falls to less than 30%.
Cement kiln exhaust is more than 70% nitrogen by volume.
The concentration of CO2 in cement kiln exhaust could be increased by firing kilns with a mixture of recycled CO2 and pure oxygen rather than air. Oxygen enrichment technology is already used to increase the output of cement kilns. Firing kilns with pure oxygen and recycled CO2 would consume much greater quantities of oxygen.
Recycling of CO2 from the kiln exhaust would be required for the preheater to continue to function.
The nitrogen ballast in the combustion air drawn into the kiln plays a vital role in the preheater lifting the feed in the gas risers between preheater cyclone collection stages. In this way the kiln exhaust would become ~95% CO2, making underground storage of the excess CO2 over and above that required for recycling more viable.
Many other process issues would need to be addressed for such a solution to be deployed. Manufacturing costs would be significantly increased, but CO2 emissions would be completely eliminated. However, the point remains that the cost of carbon must rise significantly to make research into such technologies viable.
This brings us to the end of this discussion on the possibilities for reduction of CO2 emissions associated with cement manufacture. In the next session we discuss the emissions of the other oxide of carbon, CO.
CO and volatile organic carbon compounds (VOCs).
In session 2.8 we saw that mixing of the combustion air and fuel in a cement kiln flame relies on jet entrainment of the secondary combustion air into the flame.
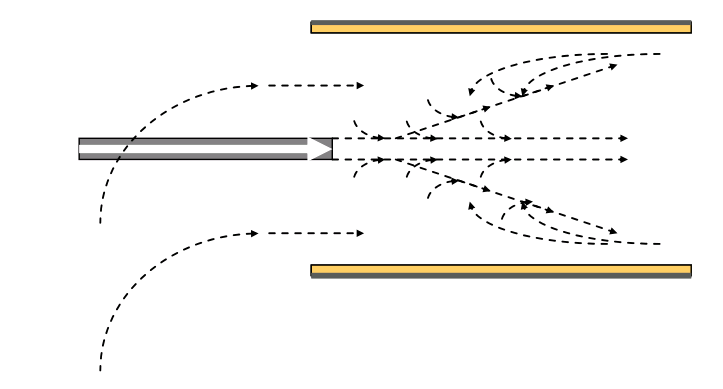
With a recirculating flame oxidising burning conditions can be guaranteed with a small excess of air above that required for combustion, as little as 1~2% oxygen at the kiln inlet.
Without recirculation the mixing of secondary air and fuel is inadequate and reducing burning conditions and CO can be present at 4~5% oxygen at the kiln inlet.
Combustion of hydrocarbon fuels in a cement kiln flame takes place by a series of free radical reactions in the gas phase.
At the temperatures in the flame the bonds in the hydrocarbon molecules in the fuel are broken to yield free radicals with unpaired electrons. These free radicals then react very quickly in a series of chain reactions. The following reactions take place in the cement kiln flame:
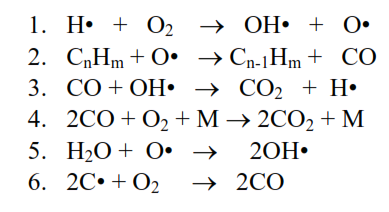
Reaction 1 is critical as it produces the OH• radicals that react with CO formed in reactions 2 and 6. Sufficient oxygen must be available in the flame for reaction 1. If not then CO and reducing conditions will be present.
Cannot rely on reaction 4 to consume CO as it is 100x slower than reaction 3 and is not significant in avoiding reduction.
With a recirculating flame and some excess oxygen at the kiln inlet then good mixing can be guaranteed and CO should not be formed in the main burner flame, provided that there is consistent delivery of fuel to the burner. An accurate and consistent feeder, plus correctly designed storage and pneumatic delivery lines are critical for solid fuels. For fuel oils the heating system to reduce viscosity and the atomisation of the fuel at the burner tip are critical.
A consistent flow of fuel, matched to the requirements of the process is the primary requirement.
Feeding discrete slugs of fuel into the kiln such as the burning of whole motor tyres violates this requirement. Increased incidence of CO in the kiln exit gases is inevitable when whole motor tyres are being burnt.
Monitoring and minimising CO in the kiln exhaust gases is particularly critical when electrostatic precipitators are used for exhaust gas dedusting. The presence of CO is used as an indicator of incomplete combustion of the fuel in the kiln and the possible presence of volatile organic compounds in the exhaust gas.
If present, these volatile organic compounds can cause an explosion in the precipitator, therefore, the high voltage is switched off the precipitator to avoid a possible explosion, resulting in a period of high dust emission while the gases are exhausted without dedusting.
Many of these precipitator trips are unnecessary, detection of CO is subject to a time lag of ~20 seconds. The high voltage has to be switched off the precipitator at CO concentrations much lower than strictly necessary as by the time the CO is detected the gases will already be in the precipitator.
When alternative fuels are burnt on a cement kiln demanding limits are placed on the CO emissions, again as an indicator of complete burnout of the fuels in the kiln. This use of CO as an indicator of complete combustion of the fuel is defeated if the kiln feed contains any organic components. The kiln exhaust gases will then contain CO, irrespective of whether the fuel has been completely burned in the kiln or not.
With organic components in the kiln feed the exhaust gases will contain CO, irrespective of whether the fuel is completely burned in the kiln.
Volatile organic compounds begin to be liberated from organics in the feed as soon as the feed enters the preheater of a cement kiln, reaching a peak at 350~400°C.
Above 350°C the volatile organic compounds are oxidised to CO, provided that 3% oxygen is present in the gases (as it would be expected to be).
CO formed by this combustion of organic components in the feed is not oxidised to CO2 until the temperature rises to above 680°C. Complete oxidation of carbon to CO2 below 680°C is inhibited by the formation of hydroperoxyl radicals rather than hydroxide radicals. Below 680°C hydroperoxyl radicals are formed:
H• + O2 → OOH•
These peroxyl radicals then react with further hydrogen radicals to form hydroxide radicals:
H• + OOH• → OH• + OH•
but the hydroxide radicals then react with further hydroperoxyl radicals:
OH• + OOH• → H2O + O2
All the radicals are consumed and the chain reactions are terminated. Availability of hydroxide radicals to react with CO is limited and oxidation of CO to CO2 is suppressed.

The liberation of volatile organic compounds from the feed as soon as the feed enters the preheater of a cement kiln introduces the possibility of further emissions problems from cement kilns. Volatile organic compounds are themselves subject to stringent emissions limits, their presence is also one of the precursors for the possible formation of dioxins in cement kiln exhaust gases.
We will consider the remote possibilities of dioxin emissions from cement kilns in the next session of the course. However, the best way to avoid these is for any raw materials containing organic components not to be included in kiln feed, any such raw materials should be fed directly to the precalciner or kiln inlet where the temperature is above 680°C and complete oxidation to CO2 will take place.
As we have seen one of the major trends in cement manufacture is the use of alternative fuels. The range of materials used is widening to include a whole class of materials whose properties are intermediate between raw materials and fuels. They have some hydrocarbon and calorific content and also significant ash content composed of the primary oxides for cement manufacture, SiO2, Al2O3, Fe2O3 and CaO. Paper sludges,
sewage sludges and rice husks are all examples of these alternative raw materials/fuels. In the case of sludges they also contain significant moisture content and therefore have negative calorific content combined with hydrocarbon content. All need to be fed to the precalciner or kiln inlet to avoid the liberation of volatile organic carbon compounds in the preheater.
Volatile organic carbon compounds are themselves subject to stringent control limits, because together with NOx they are one of the precursors of urban smog formation.
We will be devoting an entire session to NOx formation and abatement of their emissions in cement kiln exhaust gases. However, before closing the subject of volatile organic carbon compounds we need to briefly talk about their detection.
The US EPA considers any gaseous organic compound that undergoes photochemical reactions to form smog to be a volatile organic compound (VOC). This link with photochemical reactivity eliminates some organic compounds that have been shown to have neglible such activity; methane, ethane, acetone, chlorinated compounds, chlorofluorocarbons, hydrofluoro- or hydrochlorofluorocarbons. However, most cement plants measure total hydrocarbons (THCs) because it is difficult to measure VOCs. A flame ionisation detector is used that cannot differentiate between organic compounds that show photochemical reactivity. This means that cement factories that are measuring THCs are overestimating their VOC emissions.
That concludes this session of the course where we have been discussing the sources of and solutions to CO and volatile organic carbon emissions from cement kilns. In the next session we will discuss the special case of dioxin and furan emissions from cement kilns.
Dioxins and furans.
At the outset of this session we should reiterate that the likelihood of dioxin emissions from cement kilns is remote, a minimum of three preconditions must be met for their formation.
The issue of dioxin and furan emissions has arisen from the trend to burning alternative fuels in cement kilns.
Municipal waste incinerators have been identified as point sources of dioxin and furan emissions. Cement kilns burning such alternative fuels are in direct competition with these incinerators for the fees to burn such fuels. The operators of waste incinerators therefore argue that cement kilns should be subject to the same scrutiny and emissions limits as incinerators, despite cement kilns having multiple advantages over incinerators for the disposal of such wastes. However, based on the UK Environment Agency data for 1999 five UK cement sites were in the top 30 sources of dioxin emissions.
The highest dioxin emission from a cement site was 1.5 grams per year from cement factories producing ~500,000 tonnes of clinker per year. Despite these low levels of emission, dioxins are one the “dirty dozen” chemicals targeted for global phase-out or severe reduction under the persistent organic pollutants (POPs) international treaty. Under the treaty comprehensive abatement measures have to be taken to eliminate these dioxin emissions.
The reason dioxins and furans have been targeted under the POPs treaty is that they are carcinogenic, mutagenic and teratogenic – they may be cancer forming, may cause generic mutations, or may cause modification of foetal tissue. Once emitted into the environment they are also persistent, they are poorly soluble in water, not very volatile, and adsorb strongly to particles and surfaces. They are stable in the environment and accumulate in the fatty tissues of animals and humans.
There are 75 dioxins, compounds similar to dichlorobenzo dioxin (CDD), and 135 furans, compounds similar to dichlorobenzofuran. They are polychlorinated dibenzodioxins
(PCDDs) and polychlorinated dibenzofurans (PCDFs).
The best known examples are tetrachlorodibenzodioxin (TCDD):
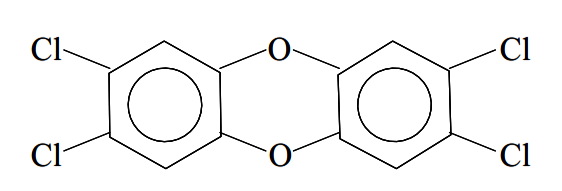
and tetrachlorodibenzofuran (TCDF):
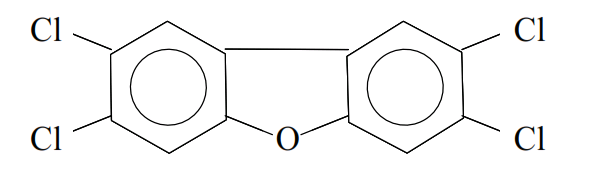
There are two mechanisms for the formation of dioxins and furans in thermal processes.
Homogenous gas phase pyrolytic reactions of chlorinated organic compounds in the temperature range 200~800°C.
Combustion of fuels in both the main burner and precalciner of a cement kiln is at temperatures above this range, however, in precalciners or secondary burners in the preheater only just above this temperature range.
Halogenated hydrocarbons might not be fully decomposed when fired in precalciners or secondary burners, therefore such fuels should only be fired in the main burner of a cement kiln where dioxin formation by this mechanism cannot take place.
A greater potential concern is the chlorination of non-chlorinated organic compounds by inorganic chloride compounds (HCl or NaCl) via a suitable transition metal catalyst. So-called “de Novo” synthesis of dioxins. This can take place in the temperature range 200~450°C by heterogeneous reaction between combustion gases and particle surfaces.
There are therefore three precursors required for dioxin formation in cement kiln exhaust gases by “de Novo” synthesis…….
Firstly the presence of unburnt hydrocarbons in the exhaust gas, which in themselves need to be avoided because of their potential for smog formation. As discussed in the previous session this is best avoided by eliminating any raw materials containing organic compounds from the kiln feed, such materials should be delivered directly to precalciners or the kiln inlet at the base of the preheater.
Secondly there must be inorganic chloride compounds (HCl or NaCl) in the kiln exhaust gases. We have already seen in sessions 1.7 and 1.8 that chlorides do build up in an external cycle between the preheater and the exhaust gas cleaning equipment, this cannot be avoided.
Thirdly there must be the presence of a suitable transition metal catalyst, usually copper as CuCl or CuCl2, a metal that might well be present in some of the process equipment.
Avoiding the presence of unburnt hydrocarbons in the exhaust gas is therefore the best defense against dioxin formation by “de Novo” synthesis, backed up by ensuring that the exhaust gases are rapidly cooled through the temperature window of 450~200°C in which “de Novo” synthesis of dioxins can take place.
This requirement for rapid cooling of the exhaust gases through this temperature window is worth thinking about. Concerns regarding “de Novo” synthesis of dioxins are a relatively new development, the process design of many kilns will not have taken this into consideration.
When electrostatic precipitators are used for exhaust gas dedusting the gases are usually cooled to below 200°C by evaporative cooling with water in a conditioning tower. Hot dry electrostatic precipitators, air-to-exhaust gas heat exchangers, or alkali bypasses might involve the exhaust gases being more slowly cooled through the critical temperature window.
These concerns should not be taken out of context. Dioxin formation in cement kiln exhaust is an extremely unlikely event, which can be avoided by complete combustion of organic compounds in fuels and feed within the process.
If dioxins are present in the exhaust gases from a cement kiln then very expensive “end-of-pipe” solutions need to be applied, cleaning the exhaust gases of dioxins prior to their exhaust from the process. This can be achieved in an activated carbon filter, or by catalytic oxidation on a vanadium or tungsten oxide catalyst supported by an alumina or titania substrate. The need for the application of such expensive technologies can be avoided by process design.
NOx formation and avoidance.
Limits on the emissions of the gaseous oxides of nitrogen from cement kilns are being lowered around the world and complying with these limits is one of the major challenges faced by the cement industry.
NOx is a generic term used to represent all the oxides of nitrogen:
NO, nitrogen monoxide or nitric oxide.
NO2, nitrogen dioxide.
N2O, nitrous oxide or laughing gas.
N2O4, nitrogen tetroxide and other less common NxOy compounds.
The temperatures in the burning zone of a cement kiln, coupled with the excess oxygen present and the long residence time favour the formation of NO. This is a colourless gas that quickly oxidises to NO2 when emitted from the kiln.
These ultimate NO2 emissions are the oxide of nitrogen which present the major problem for the Earth’s environment and the reason for the tightening emission limits. NO2 in the atmosphere causes three major pollution problems, (i) acid rain, (ii) smog and (iii) ozone formation.
1- NO2 in contact with water forms nitrous acid, HNO2, and nitric acid, HNO3, both of which are highly corrosive.When rain falls through the atmosphere containing NO2 these acids are formed, which destroy vegetation and acidify streams, rivers and lakes.
2- NO2 mixed with unburnt hydrocarbon particles in the air undergo a photochemical reaction which forms smog.
3- NO2 also reacts with oxygen in the air to form ozone.
Smog and ozone levels in the lower atmosphere are closely linked.
In the cement kiln there are two main causes of NOx formation:
Oxidation of organic nitrogen in the fuel during the combustion of the fuel, so-called “fuel NOx”.
N + ½O2 → NO ↑
Oxidation of some of the atmospheric nitrogen in the combustion air at the temperatures in the cement kiln main burner flame, so-called “thermal NOx”, or Zeldovich NOx.
N2 + O2 → 2NO ↑
“Fuel NOx” will be formed wherever the fuel is fired in the kiln, therefore the only way to avoid this source of NOx emissions is to select fuels with a low organic nitrogen content. Petroleum coke, which has been widely introduced as the solid fuel for cement kilns has higher organic nitrogen content than the coal that it replaces and therefore leads to higher fuel NOx emissions. In precalciners this is the primary means of NOx formation.
“Fuel NOx” only causes background levels of NOx emission and is not the main cause of concern. The real concern is oxidation of some of the atmospheric nitrogen in the combustion air at the temperatures in the cement kiln main burner flame, so-called
“thermal NOx”, or Zeldovich NOx.
N2 + O2 → 2NO ↑
The rate of reaction between the nitrogen, N2, and oxygen, O2, in the combustion air rises exponentially with temperature. Above 1400°C in the main burner flame this cause of NOx formation predominates.
The responses to the reduction of NOx emissions from cement kilns fall into two broad categories, (i) prevention of the formation of the NOx, and (ii) mitigating or reducing the NOx concentration in the exhaust gases after it has formed.
Prevention of NOx formation can again be sub-divided into different approaches:
Fuel NOx emissions can be reduced by conditioning techniques:
- Selection of fuels with lower organic nitrogen content.
- Pre-treatment of fuels to reduce their organic nitrogen content before firing in the cement kiln. This is an expensive approach and not employed in the cement industry.
- Addition of chemicals to the fuel to suppress NOx formation, again prohibitively expensive.
Thermal NOx emissions can be reduced if the combination temperature of the clinker can be reduced, either by using raw materials of improved burnability, changing the clinker mineralogy targets to improve combinability, or the use of mineralisers.
We have seen in preceding sessions that mineralisation and production of active belite cements would also deliver energy consumption savings, increased output and lower CO2 emissions.
Thermal NOx emissions can be reduced by adjustment of the combustion conditions in the main burner flame:
- Reducing the proportion of the primary air injected into the flame through the main burner of the kiln.
The fuels are delivered into the kiln through the main burner pipe together with the primary air, where they ignite as they encounter the kiln temperature in excess of 1400°C.
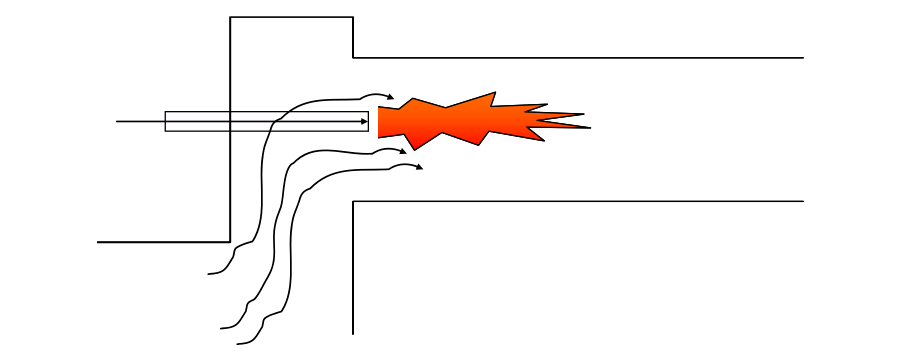
Part of the combustion air is provided by the primary air, which is blown into the kiln through the burner pipe. The remainder of the combustion air must be entrained and mixed into the flame from the preheated secondary air which is drawn into the kiln from the cooler.
We have already seen in session 2.8 that mixing of the combustion air in a cement kiln flame relies on jet entrainment of the secondary combustion air into the flame.
With a recirculating flame oxidising burning conditions can be guaranteed with a small excess of air above that required for combustion, as little as 1~2% oxygen at the kiln inlet.
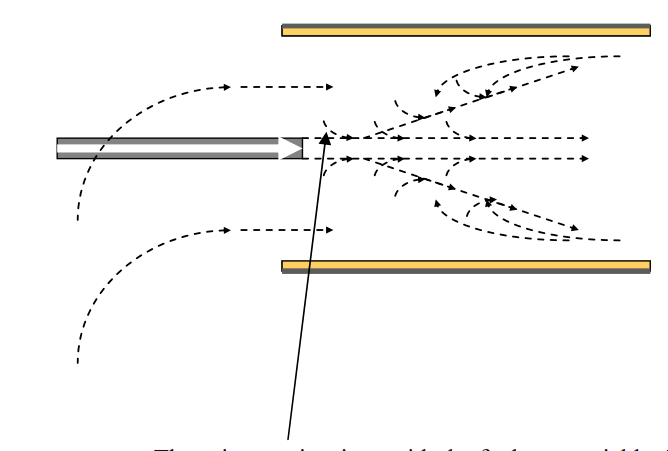
Without recirculation the mixing of secondary air and fuel is inadequate and reducing burning conditions and CO can be present at 4~5% oxygen at the kiln inlet.
The primary air mixes with the fuel very quickly. The entrainment of the secondary air is then dependent on the ratio of the primary air/fuel jet and the momentum of the secondary air. The higher the flow rate and velocity of the primary air the quicker the mixing. Recirculation is an indication that all the secondary air has been entrained into the flame and fuel/air mixing is complete.
Thermal NOx abatement by reducing the mass of the primary air relies on reducing the efficiency of the entrainment and mixing of the secondary air into the flame. This reduces the temperature in the flame and therefore the amount of thermal NOx formation. Lack of oxygen in the flame also causes the nitrogen compounds in the volatiles of the fuel to form HCN and NH3. These in turn form radicals that degrade any NOx formed in the flame by chain reactions. We will discuss these free radical chain reactions in more detail in the next session when we discuss staged combustion in precalciners.
Another strategy is to inject water or other inert liquids into the flame to suppress thermal NOx formation.
This reduces thermal NOx formation by two mechanisms:
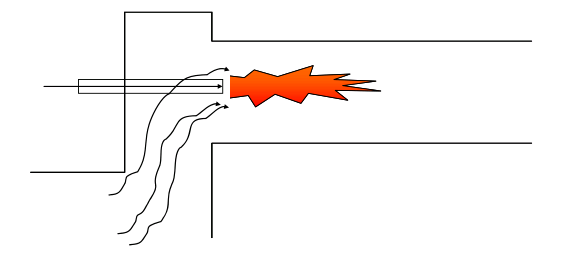
- The flame is cooled.
- Air is prevented from being mixed into the flame by the expansion of the water vapour formed.
This exclusion of air from the flame to suppress thermal NOx formation is also the strategy behind the injection of vitiated exhaust gas into the flame.
Efficient combustion in the main burner flame is reliant on the availability of oxygen in the flame. NOx formation arises from the nitrogen in the combustion air. Excluding the nitrogen in the air by enriching the combustion air with oxygen is one means of doing this. However, enrichment to 80% oxygen in air is required to achieve major reductions in thermal NOx formation.
These combustion adjustment strategies all rely on reducing the efficiency of the combustion in the main burner flame and the temperature in the flame, but we have already seen in session 2.8 that this results in the problems of poor clinker quality, and in session 1.9 that the volatility of SO3 is increased with all the attendant operational problems that that can cause. Increasingly other strategies to combat thermal NOx emissions have had to be developed.
A number of American cement companies have used oxygen enrichment to reduce the volume of exhaust gas at the exhaust fan, and boost the output of their kilns. 2% oxygen enrichment reduces the amount of nitrogen ballast drawn into the kiln with the combustion air, which in turn has to be exhausted from the kiln. A number of case studies have demonstrated that the output of a modern, dry process cement kiln can be boosted by up to 15% with oxygen enrichment of the combustion air.
This combustion air enrichment with oxygen normally concentrates on the enriching the secondary or tertiary combustion air. Enrichment of the primary air with oxygen would effectively exclude some nitrogen from the main burner flame and might assist with the reduction of thermal NOx formation.
Costs for on-site oxygen generation equipment are reducing.
Primary air oxygen enrichment technology presents a possible opportunity to both boost the output of cement kilns and improve clinker quality by intensifying the combustion ands temperature of the main burner flame, while at the same time reducing NOx formation by exclusion of nitrogen from the main burner flame.
However, oxygen enrichment of primary air is not currently being pursued for reduction of NOx emissions. The technologies that are being deployed are those that reduce the NOx content in the exhaust gases after it has formed. Those are the technologies we will discuss in the next session of the course.
NOx abatement.
In the preceding session we discussed technologies to reduce the formation of NOx in the main burner flame of a cement kiln, and saw that these can compromise the efficiency of combustion of the fuel and the quality of the clinker produced.
The second approach to NOx emission control is post-combustion mitigation or reduction of NOx after it has formed. This can be carried out either within the process, or could be carried out at the end of the clinker burning process before the kiln exhaust gases are emitted to atmosphere.
These latter “end-of-pipe” solutions are very expensive to install and operate, but may eventually be required to meet tightening emissions regulations.
Trails have taken place at various European cement factories with the “end-of-pipe” Selective Catalytic Reduction of NOx (SCR). There is a body of opinion that this represents “BAT”, best available technology.
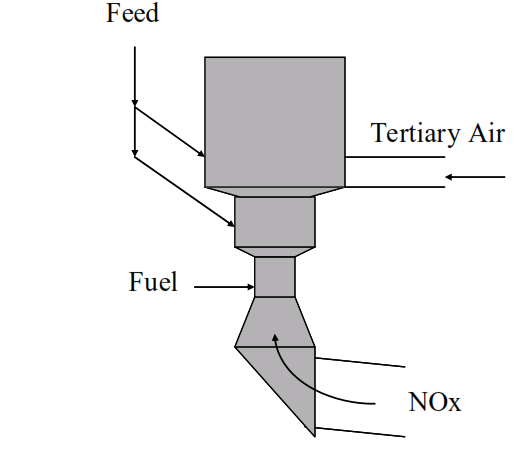
Kilns fitted with tertiary air ducts and precalciners can be designed or modified to reduce NOx generated in the main burner by the technique of staged combustion.
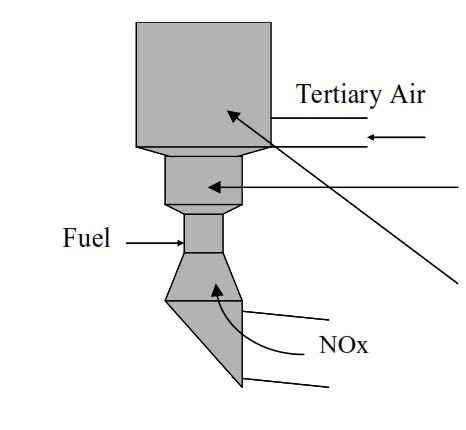
Fuel is delivered to the
precalciner below the inlet of tertiary air…
creating a reducing zone where there is insufficient oxygen available to fully combust the fuel.
After introduction of the tertiary air an oxidising zone is created in the main body of the precalciner allowing completion of the combustion of the fuel.
NOx generated in the main burner of the kiln is degraded in the reducing zone of the calciner.
Simplistically the degradation of NOx in the presence of carbon monoxide, CO, is represented by the following equation:
NO + CO → ½N2 ↑ + CO2↑
This reaction does take place, but requires the presence of kiln feed material to catalyse the reaction, and is inhibited by the presence of oxygen.
Some of the feed from the upper stages of the preheater is directed to the reduction zone in order to catalyse this reaction.
Oxygen inhibition means that the reaction ceases after the introduction of tertiary air into the precalciner. Oxygen sensitivity means the position of the tertiary air damper and the oxygen content of the kiln exit gases can also affect NOx degradation efficiency.
However, the predominant mode of NOx degradation via staged combustion in precalciners is through gas phase free radicals liberated from the volatiles in the fuel. Fuels with high volatile content are therefore the most effective at reducing NOx by staged combustion in precalciners.
Petroleum coke is therefore particularly unsuitable for firing in these low NOx calciners:
- Pet coke has higher nitrogen content than coal, generating more fuel NOx.
- Pet coke is very low in volatiles.
- Pet coke needs high oxygen content to combust.
In the absence of oxygen the nitrogen compounds in the fuel will volatilise to form HCN and NH3, which react spontaneously through chain reactions of radicals resulting in the breakdown of NOx.
NH3 is the most effective volatile nitrogen product for NOx degradation via the formation of the NH2● radical.
NH3 → NH2●+ NO → N2 + H2O
Fuels which volatilise to yield more NH3 than HCN are therefore also more effective at NOx degradation. This is thought to be the explanation for the effectiveness of animal meal at NOx degradation.
The rate of destruction of NOx by fuel nitrogen volatiles also rises with temperature. The amount of feed directed to the reduction zone is used to regulate the temperature.
In summary the pros and cons of staged combustion are as follows:
- The technique of staged combustion of fuel is only applicable to air-separate precalciner kilns, i.e. those with a tertiary duct.
- Effectiveness rises with fuel volatile content.
- Effectiveness rises with NH3/HCN ratio of volatiles.
- Effectiveness reduces with oxygen content of kiln exit gases.
For kilns without precalciners a form of staged combustion can be provided by burning alternative fuels in lump form so that they generate CO due to local shortage of oxygen at their point of combustion.
Burning alternative fuels, such as tyres, at the base of the preheater, or behind the burning zone in long kilns is successful in reducing NOx levels for the same reasons as staged combustion.
The role of the NH2● radical in the degradation of NOx is the technology involved in Selective Non-Catalytic Reduction of NOx (SNCR), which has been applied on some cement kilns in Europe for a number of years and is now being applied to some US kilns.
Ammonia water is injected into the gas stream at the base of the preheater.
The ammonia water vapourises to yield ammonia and water which reacts with NOx via the following overall reaction.
2NH4OH → 2NH3 + 2H2O + 2NO + ½O2 → 2N2 + 5H2O
NOx degradation is again via the formation of the NH2● radical by reaction between NH3 and the hydroxy radical and subsequent reaction of the NH2● radical with NO.
NH3 + OH● → H2O + NH2●+ NO → N2 + 2H2O
The degradation of NOx by reaction with ammonia reaches a pronounced temperature peak.
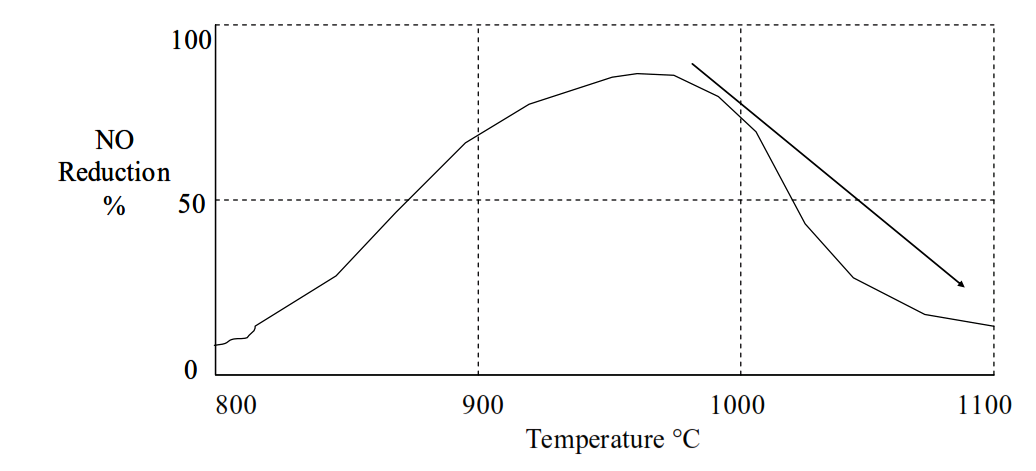
Above 980°C the NH3 reacts with oxygen to form NOx and reduce the efficiency of NO reduction:
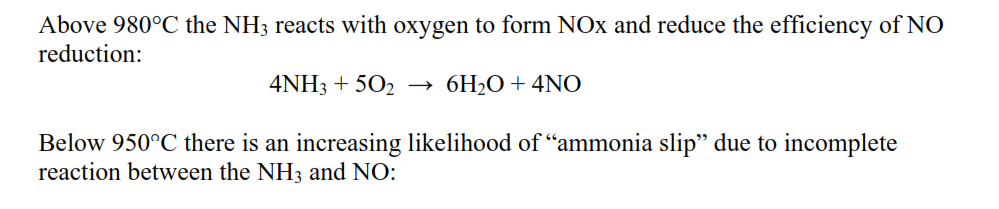
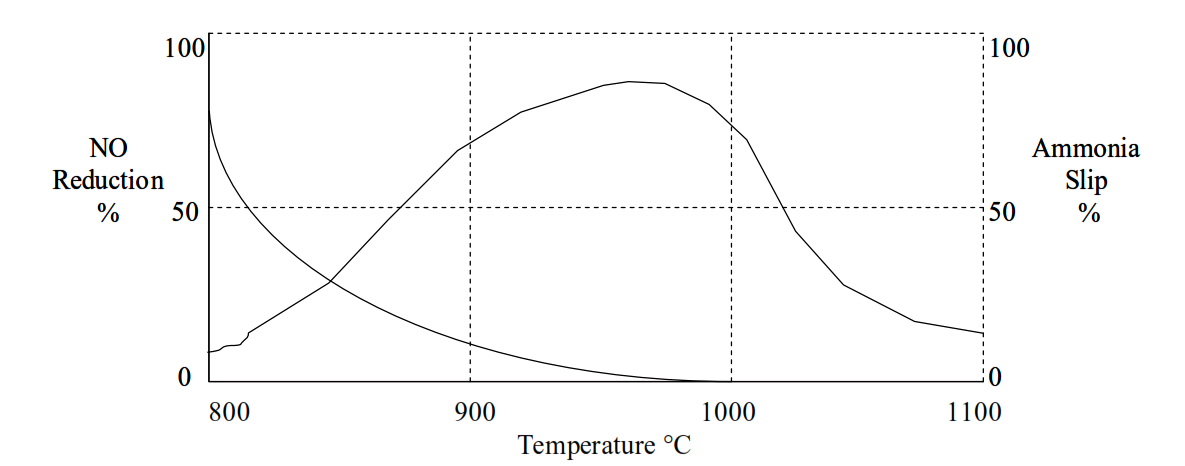
This “ammonia slip” can lead to increased emissions of ammonia, detached plumes and “blue haze” from cement kiln chimneys (these emissions will be covered in a later session of the course).
Whether the kiln is being operated in compound operation with the raw mill, or direct operation to the exhaust gas filters is the critical factor determining whether increased NH3 emissions will be encountered. Intense contact between kiln exhaust gas and raw materials in the raw mill leads to absorption of NH3 and no increase in NH3 emission.
The most effective temperature range for NO reduction by NH3 is higher than the temperatures of 860°C~910°C prevailing in the precalciner.
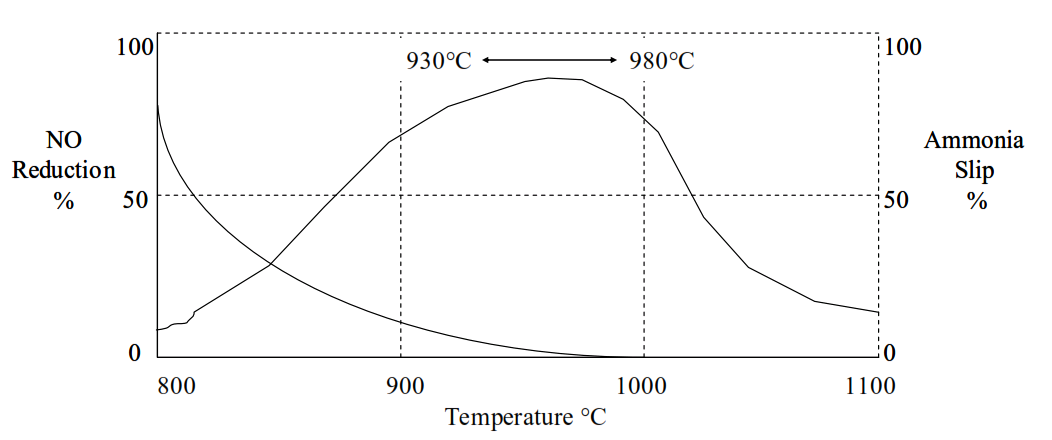
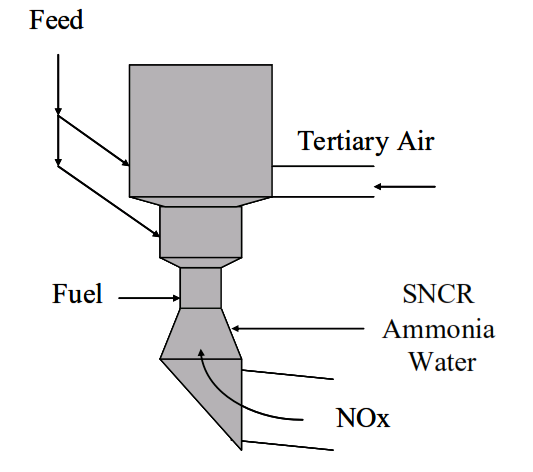
meaning that the NH3 should be injected into the gas riser from the kiln inlet to the precalciner.
The temperature at the point of injection is critical to avoid either ammonia slip or oxidation of the nitrogen in the ammonia to further NOx.
The NH3 for SNCR NOx reduction can also be provided by urea tablets, or by the ammonium thiosulphate contained in photographic waste water.
40~60% NOx. reduction can be achieved at molar ratios of less than one for the amount of ammonia injected relative to the NO in the exhaust gases at the point of injection.
Mixing the NH3 with the exhaust gas is also critical to the NOx reduction. High capacity kilns with consequently higher exhaust gas volume may require multiple injection lances for the ammonia. However, use of SNCR ammonia injection in conjunction with staged combustion for NOx reduction can lead to other problems.
The overall SNCR reaction requires the presence of some oxygen:
2NH4OH → 2NH3 + 2H2O + 2NO + ½O2 → 2N2 + 5H2O
Staged combustion is designed to generate a reducing atmosphere for NOx reduction.
With both SNCR and staged combustion there is the possibility of increased CO emissions from the kiln. Both NOx reduction techniques rely on the formation of the NH2● radical for their effectiveness:
NH2●+ NO → N2 + H2O
Formation of the NH2● radical relies on the reaction between the hydroxyl combustion radical and NH3.
NH3 + OH● → H2O + NH2●
The completion combustion of carbon to CO2 is also reliant on the reaction between the combustion hydroxyl radical and CO:
CO + OH● → CO2 + H●
NH3 therefore competes with CO for the available hydroxyl radicals, and reacts much more quickly.
The net result of these competing reactions is that both CO burn-out and NOx degradation are compromised and higher CO emissions are likely.
The latest environmental regulations in Germany require NOx emissions of less than 500 mg/m3, expressed as NO2, in dry gas at standard temperature and pressure, corrected to 10% oxygen. The EU Best Practice reference document (BREF) considers best practice to lie between 200~500 mg/m3. Cement companies can expect that emissions regulations will tighten towards this best practice.
The EU BREF document considers that the following NOx emission levels are achievable with the different NOx reduction techniques (from initial emissions of 1300~2000 mg/m3):
1- Primary measures: 500 mg/Nm3
Process control optimisation. Raw material and fuel selection. Improved fuel firing techniques.
These primary measures are a pre-requisite for control of NOx emissions.
Kiln feed burnability or clinker combinability may be reasons why these levels are not achieved with these primary measures. Also the process design of the kiln.

Historical experience suggests that cement manufacturers will eventually have to move towards SCR, probably in combination with some of these other techniques.
SO2 emissions and their abatement.
In session 3.5 we saw that formation of acid rain is one the primary reasons to reduce NOx emissions from cement kilns. This is also the main driver behind tightening regulations on SO2 emissions from cement kilns.
Cement companies operating in the European Union and North America are gradually applying expensive technologies to reduce SO2 emissions.
Sulphur enters cement kilns in either the raw materials or the fuels used to fire the kilns. However, the principal cause of SO2 emissions from cement kilns is the presence of sulphides in the raw materials, rather than sulphur compounds in the fuels.
Sulphur compounds contained in the fuels used to fire cement kilns do not usually result in SO2 emissions in the kiln exhaust.
Sulphur compounds in the fuels are oxidised to SO2 by the combustion reactions in the kiln or precalciner.
S + O2 → SO2
In precalciners the acidic SO2 immediately encounters alkaline CaO and forms calcium sulphite and sulphate…
SO2 + CaO → CaSO3 + ½O2 → CaSO4
which is then carried back into the kiln with the feed building up into a sulphate cycle in the kiln.
SO2 from the main burner comes into intimate contact with alkaline CaO in the lower stages of preheaters and again forms calcium sulphite and sulphate.
SO2 + CaO → CaSO3 + ½O2 → CaSO4
SO2 from fuels is therefore completely retained in the process.
In long kilns there is not the same degree of intimate contact between SO2 and alkaline CaO as in preheaters and therefore fuel sulphur compounds can result in SO2 emissions.
As we saw in session 1.7 sulphides in the kiln feed will oxidise in the upper stages of the preheater and are lost as gases (SO2) in the preheater exhaust gas.
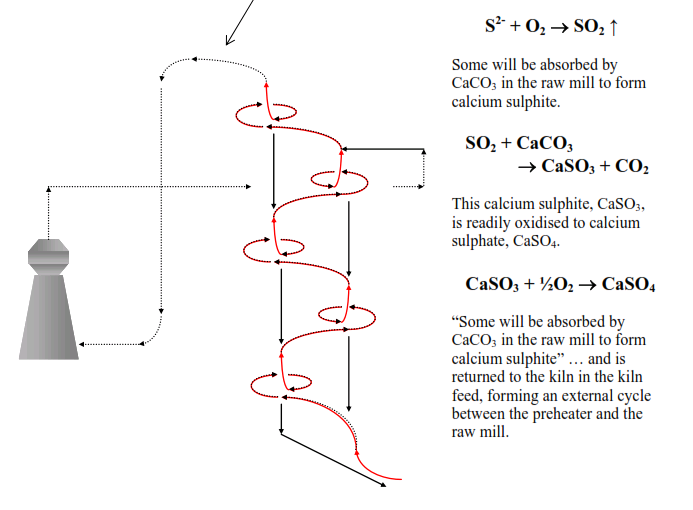
However, adsorption of SO2 in the raw mill is 80% at best. Adsorption rate falls off if the moisture content of the material in the mill is below 15%.
When the raw mill is stopped and the kiln exhaust gases pass directly to the dedusting equipment the adsorption of SO2 is drastically reduced and emissions rise dramatically. These are the circumstances that demand deployment of expensive SO2 emission abatement technologies. Many cement factories do not have sulphides in their raw materials and do not face these SO2 emission problems.
For those cement factories that do have sulphides in their raw materials the best solution is to feed these materials directly to the precalciner, the same solution as for raw materials containing organic carbon compounds.
SO2 abatement technologies for cement kilns fall into two broad categories – in process or ex-process solutions.
Ex-process solutions are also referred to as “end-of pipe” solutions – the SO2 content of the exhaust gases is reduced at the end of the process.
In-process solutions are generally less expensive to install or operate than ex-process solutions, but are only applicable in certain situations and for limited levels of SO2 emission.
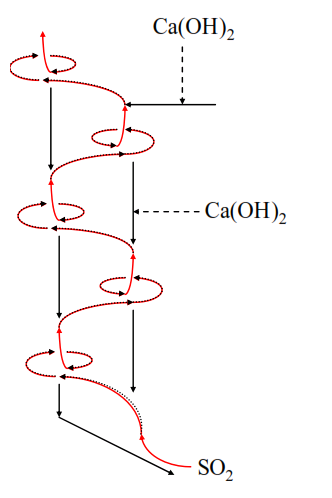
Slaked lime, (Ca(OH)2, adsorbent addition is the most commonly applied in-process solution.
The slaked lime can be added to kiln feed, or injected into the preheater.
Slaked lime does not have a high reactivity therefore a molar ratio of 3~6 for Ca(OH)2/SO2 is required to achieve 60~80% SO2 emission reduction.
If SO2 emissions are above 1200 mg/m3 the amounts of slaked lime required and the attendant costs are likely to be prohibitive. However, the addition of slaked lime can be applied intermittently to reduce peak SO2 emissions, such as when the raw mill is stopped.
Envirocare have patented a method of reducing SO2 emissions that involves injection of micro-fine lime into the conditioning tower water.
Conditioning tower water injection rates rise when raw mills are stopped and SO2 emissions are likely to rise therefore this solution matches the SO2 emission reduction requirements of the process.
However, high wear rates on the conditioning tower water injection nozzles have been reported, more information on this technique is required.
If SO2 emissions are above 1500 mg/m3 then an end-of-pipe solution becomes the most economic solution. These involve some type of scrubber to clean the exhaust gases of SO2 before discharging to atmosphere.
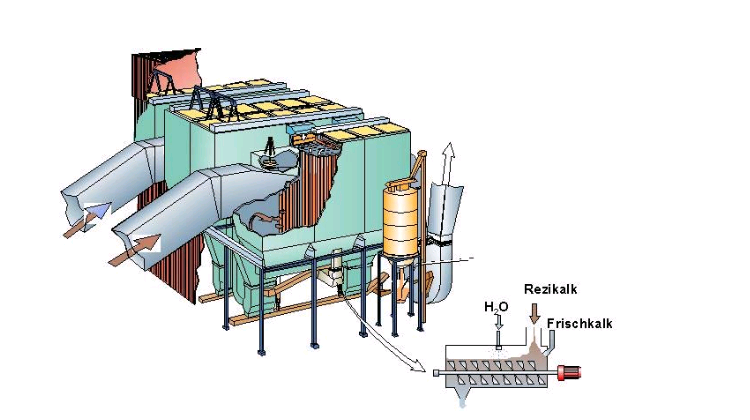
The Untervaz factory of Holcim in Switzerland has installed a dry scrubber supplied by Lurgi.
A venturi reactor column produces as fluidised bed of slaked lime and kiln feed, resulting in intimate contact between SO2 and adsorbent at close to the dew point temperature with long residence time resulting in up to 90% reduction of SO2 emissions.
The gases exiting the venturi are cleaned of adsorbent in an electrostatic precipitator, with the collected adsorbent partially returned to the column and partially directed to the kiln inlet, where all sulphur compounds will be retained in the process and discharged in the clinker.
Wet scrubbers are more commonly applied for reducing SO2 emissions in power station and cement factory exhausts (although this is not yet common in cement manufacturing).
SO2 is absorbed in a liquid slurry containing finely ground limestone, CaCO3, that is sprayed in counter-current to the gas flow.
Calcium sulphite, CaSO3, is formed and the slurry is collected in a recycle tank at the bottom of the scrubber, where the sulphite is oxidised to gypsum, CaSO4.2H2O. After separation the gypsum is used in cement milling and the water is recycled to the spray tower.
SO2 emissions are reduced by up to 90% to less than 200 mg/m3, from peak values in excess of 2000 mg/m3 at the inlet.
The cleaned gases have to be reheated to above their dew point before exhausting to atmosphere, this can achieved by mixing with cooler exhaust air.
An alternative to wet or dry scrubbers are activated carbon filters such as installed at the Siggenthal factory of Holcim, Switzerland.
Deactivated coke is extracted from the bottom of the filter and activated coke is charged from the top. The deactivated coke is then burnt as an alternative fuel on the kiln.
At Siggenthal SO2 emissions are reduced by 90% to less than 50 mg/m3 from maximum at the inlet of 600 mg/m3.
Activated carbon filters will not only clean SO2 from the exhaust gases, volatile organic compounds (VOCs), dioxins, metals, ammonia and ammonium compounds, HCl and HF will also be removed in addition to any residual dust. This is in fact a general advantage of these “end-of-pipe” emission reduction technologies. A wet scrubber will also significantly reduce HCl, metal, ammonia and residual dust emissions. Dedusting of the gases prior to these filters is a requirement to prevent blinding of the filters, however there are “end-of-pipe” technologies available that combine dedusting with SO2 removal.
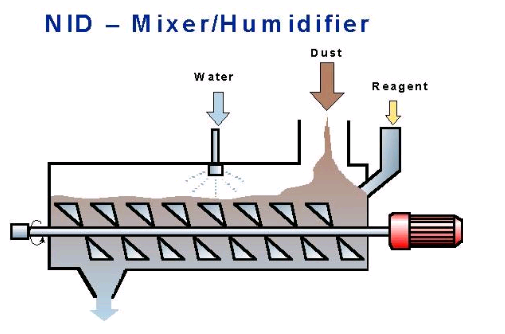
Filsorption systems combine absorbent injection with bag filters.
An absorbent mixer/humidifier can be installed beneath a bag filter or electrostatic precipitator.
These “end-of-pipe” emission abatement technologies seem certain to find wider application in cement manufacturing as emissions regulations continue to tighten and pressure to apply “best available technology”, BAT, increases. The growing trend of burning alternative fuels invites this pressure.
The advantage of these “end-of-pipe” technologies in solving multiple potential trace emission problems from cement kiln exhausts is particularly relevant to the emissions we will discuss in the final sessions of the module 3 – plumes, blue hazes and heavy metals.
Plumes and “blue haze”.
Some cement factories suffer from intermittent, or permanent, plumes forming in the atmosphere from their kiln exhaust.
Cement kiln exhausts inevitably produce a plume of steam when the gases meet the atmosphere. We have seen that the exhaust contains water vapour from the combustion of the hydrogen in the fuel in addition to any evaporated moisture in the kiln feed and moisture dried from the raw materials in the raw mill.
The time and distance taken for this steam plume to disperse is dependent on ambient temperature and humidity.
Plumes become a problem when they persist beyond the dispersal of the steam plume, or manifest themselves as a detached plume from an exhaust stack with no apparent steam plume.
The general public perceive these as plumes of dust. However, the electrostatic precipitators or baghouses installed on modern cement kilns reduce dust emissions to below the levels that would be perceptible to the general public.
Nevertheless, such plumes can be visible with the most efficient ESPs and baghouses.
These plumes are caused by the condensation of fine particles of volatile compounds from the exhaust gases. Volatile compounds that pass through the dedusting equipment and out of the process as gases and only condense when they meet the ambient temperature of the atmosphere. These volatile compounds are usually ammonium chloride or sulphate derived from minor traces of ammonium compounds in the raw materials. Very low concentrations of these ammonium compounds can cause these detached plumes.
The particles formed are in the 0.3~0.6 micron diameter size range, which causes the highest attenuation of light and maximises the visible impact of the plume.
Ammonium compounds in the kiln feed exhibit similar behaviour to sulphides in the kiln feed.
Ammonium compounds evaporate in the upper stages of the preheater yielding ammonia, NH3.
Some of the NH3 is absorbed by CaCO3 in the raw mill and is returned to the kiln in the kiln feed.
NH3 can concentrate up to 10x the input levels in this external cycle.
When the NH3 scrubbing capacity of the raw mill is exceeded, or when the raw mill is stopped, the excess NH3 passes to the chimney and forms the plume.
Only small concentrations of NH3 and SO2 (30~50ppm) in the exhaust gases are required to form a plume.
SO2 emission abatement technologies, addition of Ca(OH)2 adsorbent to the raw mill, lime injection into conditioning tower water, dry or wet scrubbers, will be effective in eliminating (NH4)2SO4 plumes.
The intermittent appearance of the plume can arise due to the switching from direct operation to combined operation when the raw mill starts and stops.
These plumes can also be caused by ammonium chloride, NH4Cl. We saw in session 1.8 that chlorides concentrate in the lower stages of the preheater. However, some chloride escapes as gaseous HCl, with less than 100ppm being sufficient to cause an NH4Cl plume.
In a well documented case at Guadalahara in Mexico a persistent NH4Cl plume was solved by a series of process modifications designed to reduce the temperature of the gases exiting the preheater and entering the dedusting equipment, which resulted in condensing the NH4Cl before it entered the dedusting equipment.
In the next and final session of module 3 of the course we will focus on the remote possibility of heavy metal emissions from the cement manufacturing process, in particular mercury and thallium emissions, control of gas temperatures through the process is certainly a recognised means of controlling mercury emissions.
Heavy metals – mercury and thallium.
As a further consequence of burning alternative fuels in cement kilns and the generally tightening regulatory environment within which cement factories operate, heavy metal emissions have inevitably come under scrutiny.
With regard to emissions, two heavy metals; mercury, Hg, and thallium, Tl, attract the greatest attention as a consequence of their volatility and their toxicity.
Mercury is the only metal that is a liquid at room temperature, meaning that the metal and the salts of the metal have high vapour pressures and volatility.
All mercury compounds in the feed to a cement kiln will evaporate in the preheater and be drawn through the induced draft fan as gases. Mercury metal vapours are extremely toxic, meaning that emission limits are restricted to fractions of a milligram per cubic metre of cement kiln exhaust.
Mercury is only present as minute traces in cement raw materials, normally the extremely low emission limits are not approached, let alone exceeded, in cement kiln exhausts. The emission levels are so low as to be at the limits of detection. Alternative fuel burning raises the possibility of higher mercury emissions, particularly refuse derived fuels and sewage sludge.
As mercury exits the preheater as vapour the temperature regime along the gas flow path behind the induced draft fan is critical for retention of mercury within the process. The critical factor is the temperature of the gases entering the final filter. When operating in combination with drying raw materials in the raw mill the temperature can be as low as 90°C, just above the dew point of ~77°C. These low temperatures and the intimate contact with the raw mill product and dust mean that mercury compounds condense on the mill product. These condensed mercury compounds then build up into an external cycle between the preheater and the raw mill. When the raw mill is stopped the temperature entering the final filter rises and the intimate contact with material in the raw mill is lost. Mercury emissions to atmosphere therefore rise when the raw mill is stopped. These mercury emissions when the raw mill is stopped are an effective bleed on the external cycle, but can also exceed emission limits.
The solution to these mercury emissions when the raw mill is stopped is an intermittent dust purge when the raw mill is running. The level of recirculating mercury in the external cycle needs to be allowed to build up to greater than the input levels in the feed and fuels, then the dust is purged from the system for a period to bleed mercury out of the external cycle. In that way the level of mercury circulating in the external cycle is controlled and at a sufficiently low level when the raw mill stops. The emissions when the raw mill stops are lower than the emission limits. Intermittent dust purges are also the means to control the cycles of thallium, along with selection of raw materials that do not contain this toxic metal.
Thallium is almost as volatile as mercury. All the thallium inputs to a cement kiln will again evaporate in the preheater, measurements in each stage of preheaters have shown the greatest concentration of thallium chloride in the top stage cyclone. In common with mercury the volatility of thallium is such that none will exit in the clinker and cycles must be broken in other ways. Significant quantities exit the preheater in the dust and build up into an external cycle between the dust filter and preheater.
Thallium is also extremely toxic, gastro-entiritus is followed by hair loss and attack on the central nervous system and death. Vegetation loss and balding of rabbits was the first indication of the thallium emissions from the well documented case of the Lengerlich cement factory in Germany.
The most common source of thallium inputs to cement manufacturing are iron additives. At Lengerlich residues of pyrite roasting were used as an iron additive. The dust filters were highly efficient at collecting cement kiln dust, but the thallium was found to be bound to particles in the 0.2~0.8 micron size range. The dust filter only achieved 50% efficiency in collection of these thallium rich particles, between 140~200 grams of thallium per hour were emitted.
Elimination of the pyrite roasting residues from the raw mix reduced these thallium emissions by more than 99%. An intermittent dust purge is the solution if traces of thallium are present in the main limestone or clay raw materials of a cement factory. Again intermittent to allow the level of recirculating thallium in the external cycle to build up to the levels entering the kiln in the feed and fuels.
This brings us to the close of our exploration of the emissions from cement kilns, and indeed, almost to the end of the entire kiln chemistry course.
In the next and final session of the course we will review the information presented over the last six weeks, and venture some suggestions regarding the optimum ways to operate a cement kiln from a chemistry perspective.
Conclusion
After six weeks we have reached the final session of the Cemnet cement kiln chemistry course.
During the first two weeks in Module 1 we focused on issues relating to the efficiency and productivity of cement kiln operation.
During the middle two weeks in Module 2 we focused on issues relating to clinker product quality.
During the final two weeks in Module 3 we have focused on the emissions from a cement kiln.
At the outset of the course I said that we would attempt to explore the overlapping issues between these principle causes of concern when operating a cement kiln.
Also that we would try to find the optimum strategies to satisfy these sometimes conflicting objectives.
In module 1 we began with the factors that affect the burnability of the raw mix and combinability of the clinker. Without doubt any adjustment that improve kiln feed burnability or clinker combinability will reduce energy consumption and improve clinker productivity. Lower energy consumption will reduce CO2 emissions and lower burning zone temperatures will reduce NOx emissions, a positive overlap of these objectives. But reductions in lime saturation or silica modulus are likely to be detrimental for clinker quality as they reduce C3S content.
Module 1 was where we first talked about the adverse effects of combustible, organic materials in the kiln feed. These tend to lift preheater exit temperature and therefore adversely affect energy efficiency. In module 3 we saw that there are multiple reason to avoid combustible materials in the kiln feed – CO, volatile organic carbon, even possible dioxin emissions, reducing conditions might even adversely affecting quality, a situation to be avoided for many reasons.
Exotic technologies such as oxygen enrichment to boost kiln output were touched upon. Oxygen enrichment might also allow emissions to be reduced or controlled, combustion of alternative fuels would certainly be intensified, enrichment of primary air would exclude nitrogen from the flame and may result in lower NOx emissions, however, further research and trials would be required.
We spent 3 sessions talking about the details of alkali cycles in the cement kiln and external cycles between the kiln and raw mill. These external cycles can cause emissions problems, but the key is to understand them and control them. With understanding and control no adverse affects should arise for efficiency, emissions or quality. A recurrent theme was the need to balance the alkali and sulphates in the process and the clinker product, for quality, consistency and lack of variation of alkali sulphates in clinker is critical as these strongly influence early strength.
We talked about the importance of the combustion in the main burner flame for clinker quality. A sharp, hot temperature profile is required, as can be provided by the recirculatory flame. This means that NOx will be generated in the flame and technologies such as staged combustion or SNCR need to be applied to degrade this NOx in the preheater, or by the end-of-pipe SCR technology.
Burning alternative fuels is a growing trend in the cement industry. This invites stronger scrutiny on emissions and tighter emission limits, but there is no reason why burning alternative fuels should mean higher emissions, provided that the systems for delivery of the fuels to the kiln, the point of addition and the combustion are correctly designed. Indeed, biomass fuels are carbon neutral and therefore reduce emissions.
However, the technology that has the maximum positive effects on energy efficiency, productivity, emissions and clinker quality is mineralisation. Burning zone temperatures and thermal energy consumption are lowered, fan capacity is released to boost output, NOx and CO2 emissions reduce with the burning zone temperature and fuel consumption, strengths can be boosted by higher C3S, or CO2 emissions can be further reduced by producing active belite cements.
In the long term technologies such as mineralisation, active belite cements, SNCR and SCR NOx abatement, end-of-pipe scrubbers, sequestration and co-production of electrical energy and cement are likely to be adopted. A sound knowledge of cement kiln chemistry will then take on an even greater importance. We hope that this course will have helped prepare for this future.
If You Want to Download the Complete Cement Kiln Chemistry and All Complete courses and Expert Books in Cement Industry , Kindly click here NOW