Contents
To download the below and all other Useful Books and calculations Excel sheets please click here
To download the below and all other Useful Books and calculations Excel sheets please click here
Raw materials used for Cement Production

Have you ever asked your Self the Below Questions ?
what are the two raw materials used to make cement
how is cement prepared
cement raw materials percentage
name the important raw material used in manufacturing of cement
cement raw materials pdf
cement composition percentage
cement composition pdf
today in this Article I will try to Answer all these Questions and More…..
Raw materials
Minerals of natural origin as well as industrial products can be used for the production of cement. Starting materials for this purpose are mineral compounds containing the main components of cement: lime, silica, alumina and iron oxide. Seldom are these components found in the needed proportion in only one raw material. Therefore it is usually necessary to select a measured mixture of a high lime component (lime component) with a component which is lower in lime, containing however more silica, alumina and iron oxide (clay component). These two main components are generally limestone and clay or limestone and marl.
Lime component
Limestone
Calcium carbonate (CaC03) is wide spread in nature. Calcium carbonate of all geological formations qualifies for the production of Portland cement. The purest grades of limestone are calcspar (calcite) and aragonite. Calcite crystallizes hexagonally, and aragonite is rhombic. The specific gravity of calcite is 2.7 and of aragonite 2.95. A macroscopic-granular variety of calcite is marble. It would not be profitable to use marble for cement manufacture.
The most common forms of calcium carbonate, most similar to marble, are limestone and chalk. Limestone is of a predominantly fine grained crystalline structure. The hardness of limestone depends on its geological age; usually, the older the geological formation, the harder the limestone. The hardness of limestone is between 1.8 and 3.0 of the Mohs’ scale of hardness; its specific gravity is 2.6 to 2.8. Only the purest varieties of limestone are white.
Limestone usually contains admixtures of clay substance or of iron compounds, which influence its color.
Chalk
Chalk is a sedimentary rock which was formed during the Cretaceous period in geological time; geo logically it is relatively In contrast to limestone, chalk is characterized by a soft earthy texture; this property qualifies chalk as a raw material especially for the wet process of cement manufacture. Since blasting is not required for quarrying of chalk, and the crushing process can also be omitted, this kind of raw material considerably lowers the cement production costs. In some deposits the calcium carbonate content of chalk amounts to 98-99 0/o, with small admixtures of Si02, Al203, and MgC03.
In cement raw materials the lime component is generally represented up to an amount of 76-80 0/o. Therefore, the chemical and physical properties of this component are of decisive influence, when it comes to selecting a method of cement manufacturing as well as the type of production machinery.
Marl
Limestone with admixtures of silica, clay substance, and iron oxide are called marls. Marls form the transition element to the clays. Because of the wide distribution of marls, they frequently are used as raw material for cement production.
Geologically, marls are sedimentary rocks generated by simultaneous sedimentation of calcium carbonate and clay substance. The hardness of marls is lower than that of limestone; the higher the content of clay substances, the lower their hardness. Sometimes marls contain also bituminous constituents. The color of marls depends on the clay substance and ranges from yellow to grayish-black. Marls are an excellent raw material for cement manufacture, because they contain the lime and the clay component in an already homogenized condition. Calcareous marl of the chemical composition equal to that of Portland cement raw mix, are used for the manufacture of so-called natural cement; however, deposits of such raw materials are not common. As to the quantitative proportions of the lime and clay components in cement raw materials, the following classification is established:
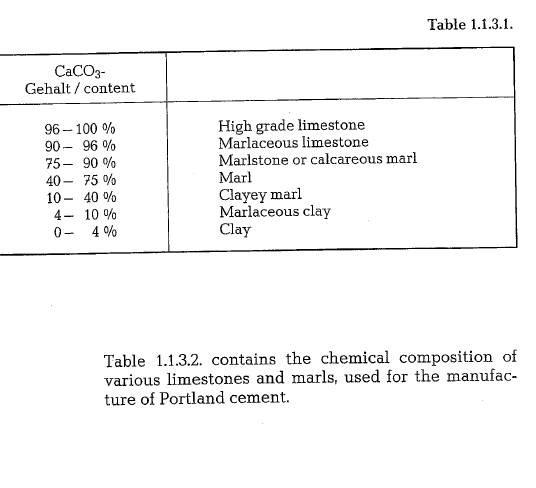
Clay component
The other important raw material for cement manufacture is clay. Clay is formed by the weathering of alkali and alkaline earth containing aluminum silicates and of their chemical conversion products, mainly feldspar and mica.
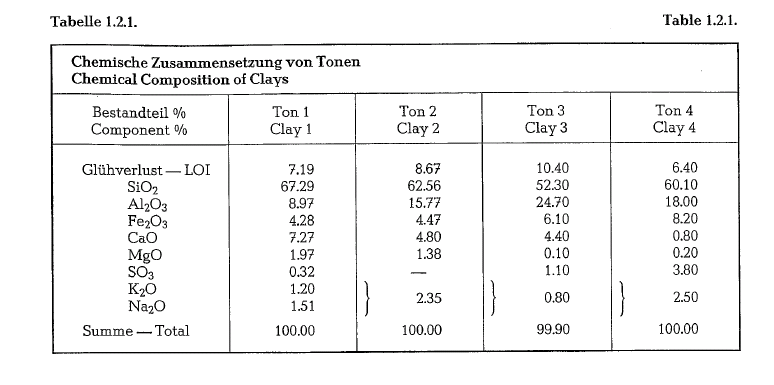
The main component of clays is formed by hydrous aluminum silicates. Clays are divided into the following mineral groups [6]:

Minerals of the kaolin group differ in content of Si02 as well as by the crystallographic structure and opti cal properties. The designation kaolinite refers to the pure kaolin mineral. Texturally, clays are fine grained; clay particles are usually under 2 microns in diameter.

The fusion point of clays is within the range of 1150 oc to 1785 oc (Seger cone No.1 to No. 35, or Pyrometric Cone Equivalent).
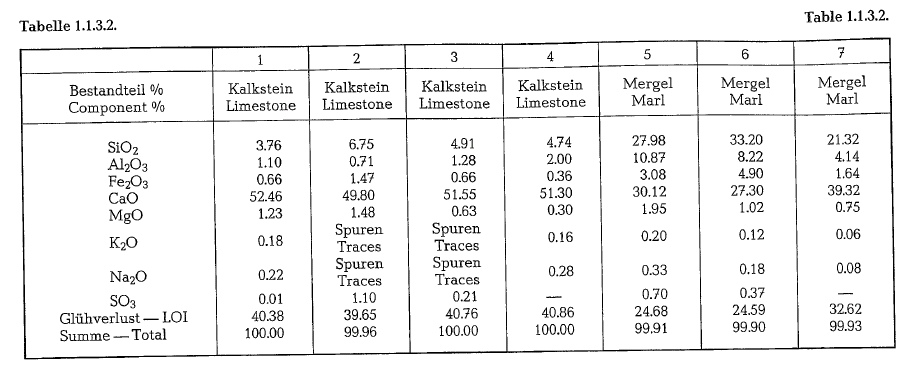
The chemical composition of clay may vary from those close to the pure clay, to that containing a considerable amount of chemical admixtures as iron hydroxide, iron sulfide, sand, calcium carbonate, etc. Iron hydroxide is the principal coloring agent in clays; also organic matter may give the clay different colors. Clays with no impurities are white. The major source of alkalies in cements is found in the argillaceous component of the raw mix.
The following tabulation shows the chemical composition of various clays, used for the manufacture of Portland cement.
Corrective ingredients
If an essential chemical component needed in the cement raw mixture is not present in the required amount, corrective ingredients are used as additives. Thus e. g. for the completion of the silica content, sand, high silica clay, diatomite, etc., are used as additives or corrective ingredients. To compensate for deficiency in iron oxide, such corrective materials as pyrite cinders, iron ore, etc., are applied.
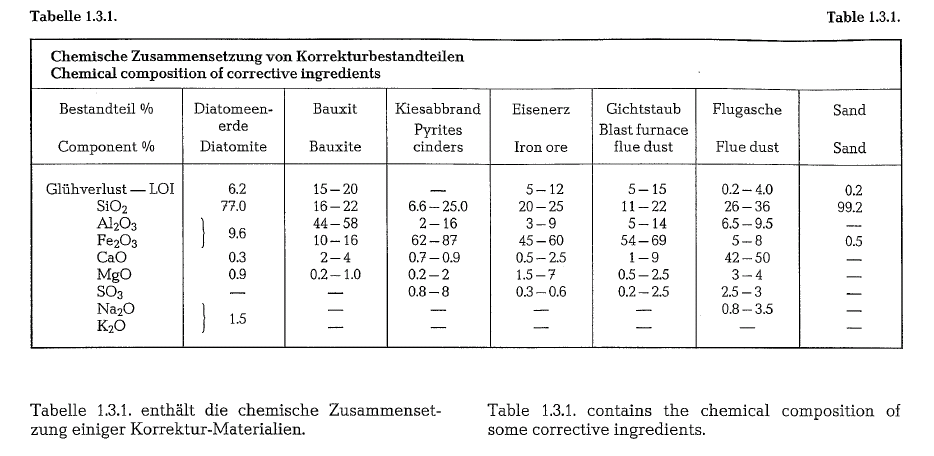
Auxiliary components of cement raw materials
Here, such components are discussed, whose quanti ties in the cement are limited either by standard spe cifications or by manufacturing experience.
Magnesium oxide
This component is combined up to 2 % by weight with the main clinker phases; beyond that amount it appears in the clinker as free MgO {periclase). Peri
clase reacts with water to Mg(OH)z: MgO + H20 =
Mg(OH)z, but this reaction proceeds slowly, while the
other hardening reactions are already concluded. Since the Mg(OH)z occupies a larger volume than the MgO and is formed on the same spot where the periclase particle is located, it can split apart the binding of the hardened cement paste, resulting in expansion cracks (magnesia expansion),
MgO appears in limestone mainly as dolomite (CaC03 · MgC03).
Also blast furnace slag sometimes contains a high amount of MgO. When selecting such slags as a replacement of clay as the argillaceous component of the raw mix, care should be taken, to keep the MgO content of the clinker within permissible limits (see example 2.5. and table 2.5.).
Alkalies
K20 and Na20 originate from such raw materials as clay and marl, where these compounds are present in fine dispersed feldspar, mica and illite particles, and to a small amount -when using coal as fuel -from the coal ash [139, 245, 7a]. In Central Europe, the K20- content represents a multiple of the Na20-content.
IN other areas as e. g. the USA, the Na20-content is pre dominant (see tables 1.1.3.2. and 1.2.1.). During the burning process in the rotary kiln, parts of the alka lies evaporate in the burning zone, causing the so-called alkali circulation
Certain concrete aggregates, appearing e. g. in some areas of the USA, Denmark and Northern Germany, contain alkali-sensitive components as e. g. opal (hydrated silica), reacting with the alkali of the cement, which under certain unfavorable conditions can result in an expansion phenomena (alkali expan sion). As is known from experience, alkali expansion can be prevented when using cement with low alkali content; in this case the total alkali, calculated as Na20 (%by weight Na20 + 0.659% K20) should not exceed 0.6 % by weight. With reference to restrictions applied in other countries [14], Germany also decided for a limit of 0.6 % Na20 equivalent, but only for Portland cement. Practical experience showed namely that for blast furnace slag cement the limit for the admissible alkali content can be higher; thus a “cement with a low active alkali content (NA cement)” with a slag component of at least 50 % can have a maximum alkali content of 0.9 %. Further, a total alkali limit of max. 2% is allowed for cement with a slag component of at least 65% [7b].
In cases in which NA-cement is required, but the alkali content of the clinker burned from the avail able raw materials exceeds the allowable limit, part of the kiln exit gases must be bypassed before entering the preheater, to remove a portion of the volatile alkalies. For particulars see 20.6.1. to 20.6.4.
Here it should be mentioned that Federal specifications SS-C-192 b, AASHO-specifications, as well as ASTM-specifications have optional alkali limits of 0.6% as Na20 in Portland cement. This means that the customer must specify that he wants alkalies under this limit, otherwise there are no limits on alkali content. The limit may be specified when the cement is to be used in concrete with aggregates that may be deleteriously reactive. But because of the difficulty of segregating low- and high-alkali cements, it has become common practice in many areas of the United States for all cements to conform to low-alkali specifications; see also [7d, 7e, 7f].
Sulfur
Sulfur appears predominantly as sulfide (pyrites and marcasite, FeS2), in almost all cement raw materials. An investigation of more than 90 German limestone deposits showed a total sulfur content (sulfide and sulfate compounds) of max. 0.16% S, and the examination of 67 clay depositis showed an average of 0.22 % S. The sulfur content of fuels varies in wide limits, from zero in natural gas, to about 3.5 % in heavy fuel oil. Coal from the Ruhr basin (W.Germany) shows an average of 1.1 % S [1]. Investigations of 21 German preheater kilns showed that the amount of sulfur introduced with the raw mix, varied in the range from 0.5 to 11 grams S03/kg clinker, and the sulfur introduced with fuel, when using extremely high sulfur fuel oil, showed a maximum of 6 grams S03/kg clinker [7a, 139].
As a result of the combustion, the sulfur of the raw mix and of the fuel evaporates in the burning zone to S02, which in the kiln atmosphere combines with alkali and oxygen, generating vaporized alkali sulfate, which condenses on raw mix particles in colder kiln zones as well as in the preheater. With the exception of a small part which is carried away by the kiln dust, the alkali sulfate returns with the kiln feed into the burning zone, and – due to its low volatility – leaves the kiln with the clinker.
If the S02-content is insufficient to combine all alkalies, the circulation of the medium volatile alkali carbonates or of the high volatile alkali chlorides begins (see section 1.4.4.). So far as the alkali carbonates are not bound by the clinker phases (see section 1.5.) they evaporate again in the burning zone.
In the preheater, an excess of S02 is able to react with the CaC03, and to return to the kiln as CaS04. Then in the burning zone, it decomposes again and thus increases the S02-circulation of the kiln gas. However, a certain part will appear in the clinker as undecomposed CaS04•
The presence of alkalies in the raw mix in excess of the amount which is already combined by the sulfur contained in the raw mix, allows the use of high sulfur fuels without emitting noteworthy quantities of S02 by the kiln exit gases. The alkali sulfate com bined by the clinker is of advantage for the early strength of the cement. On the other hand, higher sulfur contents can result in increased so2 emission with the exit gases, choking of the suspension preheater, as well as formation of kiln coating rings.
To control the setting time, cement needs a minimum amount of calcium sulfate, mostly in the form of gypsum added to the clinker. On the other hand, the maximum allowable S03-content in the cement -to prevent “sulfate expansion” -is established according to the various cement standards, between 2.5 and 4% S03. At the lowest limit imposed by cement standard specification, it could be possible that there is no sufficient scope left for an extensive sulfatizing of the alkalies.
Chloride
The chloride content of German cement raw mix fluc tuates generally between 0.01 and 0.1% by weight, sometimes rarely over 0.3% [7a]. As already mentioned above, the chloride reacts with the alkalies in the rotary kiln, forming alkali chlorides, which leave the rotary kiln with the gases, and condense in the preheater. They return into the kiln with the raw mix, but unlike the alkali sulfates, they vaporize almost completely in the burning zone. Since they condense quantitatively in the preheater, they circulate between burning zone and preheater, until increasing coating requires shut-down of kiln operation. To prevent this phenomenon, part of the kiln gases (up to 10 or 25 %), are bypassed and do not enter the pre heater. As is known from practice, bypassing of kiln gases is necessary at a chloride content of more than 0.015% Cl in the raw mix [7a].
Previously, calcium chloride was added to high early strength cements to boost the initial strength of the cement. However, it appeared that chloride promotes corrosion of steel, which had a detrimental effect on the tension wires in prestressed concrete. Therefore, the revised German cement standard specification DIN 1164 of 1970, does not allow addition of chloride to the cement. However, traces of chlorides to a max imum of 0.1 % Cl are admitted, if they come from the raw material [7c, 139].
fluoride
The fluoride content of conventional cement raw materials fluctuates within the limits of 03 and 0.08 %. In contradistinction to the chlorides, the fluorides do not volatilize easily, and thus do not disturb operations, since they do not circulate in the kiln system. The former practice of adding up to approximately 1 % of calcium fluoride to the raw mix, to facilitate clinker burning, is now in most cases abandoned, since the binding of lime can be achieved by a higher degree of homogenizing and finer grinding of the raw mix.
phosphorus
The phosphorus content of the commonly used natural cement raw materials is very low; in Ger many, the P205-content of the clinker is within the li;mits of about 05 and 0.25 % [139]. In cases where raw materials with a higher phosphorus content are used (e. g. limestone with a high P205-content, or industrial wastes from phosphorus production), an admissible limit of about 2.5% P205 in the clinker is quoted [12b]. However, it shoud be noticed that some times P205-contents as low as 0.5 0/o can already cause a considerable decrease in strength, especially in the early strength of the cement [12c].
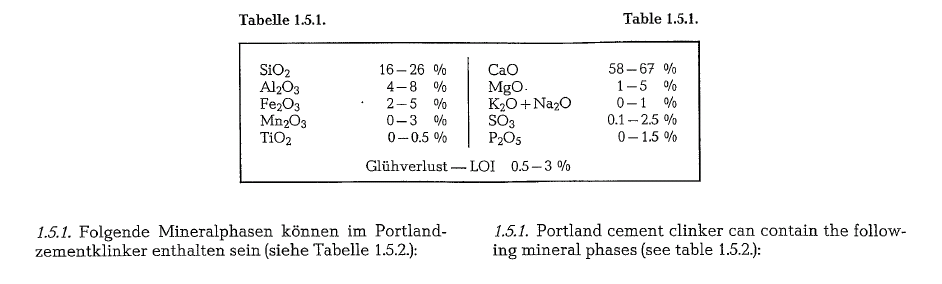
Mineral phases of the Portland cement clinker
Portland cement clinker contains approximately the following chemical constituents:
Clinker minerals
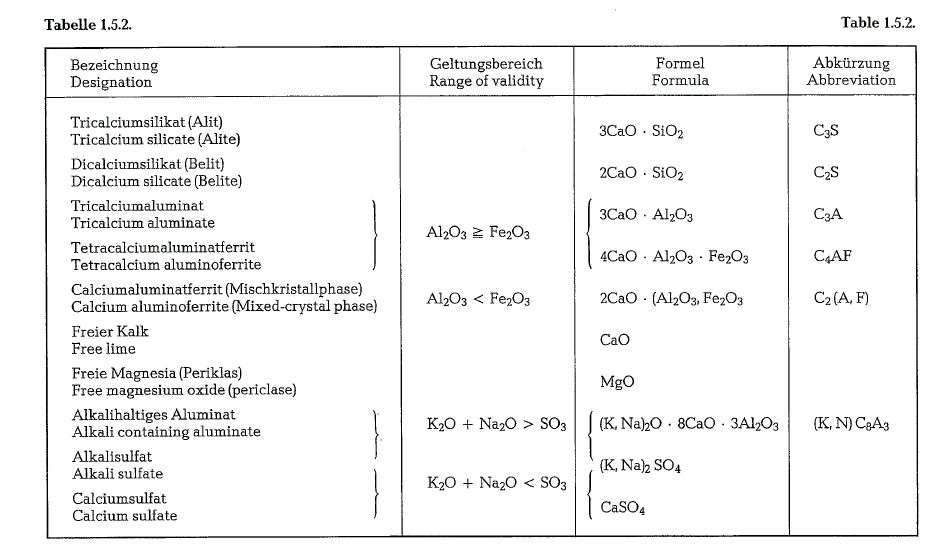
clinker Minerals are not pure compounds but mixed crystal phases, which contain constituents of other phases in small amounts as combinations of mixed crystals; these crystals contain also the remaining chemical admixtures of the clinker, which are not able to form separate phases. To characterize the difference between the pure compounds and the clinker minerals, the designations for the main clinker components, namely alite for C3S, and belite for C2S were retained in practical use. Tornebohm coined these designations in 1897, when it was possible to identify these minerals microscopically, but when their chemical composition was not known.
Alite (C3S)
Alite (C3S) is the prevalent clinker mineral, determining its property of Six modifica tions of C3S are known, of which only 2 high tempera ture modifications appear in cement clinker; these modifications are stabilized by insertion of unrelated atoms [12a, 139].
Belite
Belite is mainly the -modification of C2 At sintering temperature of the clinker, if this tempera ture is above 1420 oe, a-C2S is generated; if this temperature is below 1420 oe, a’-C2S is formed. This modification changes during clinker cooling at about 670 oc into the metastable -C2S. At further slow cooling, inversion into the hydraulically inactive y-modification takes place. The transition from the-C2S into the y-modification is accompanied by an increase in volume of about 10 %, whereby the clinker disintegrates. Fast cooling and chemical mixtures which are almost always present in the belite, prevent the transition from the -modification into the detrimental and hydraulically inactive y-modification.
Belite hardens considerably slower than alite; how ever, after a longer period of time it attains the same strength.
alumina
If the clinker contains less alumina than iron oxide (calculated in moles), then both constituents combine with lime, forming calcium aluminoferrite (see table 1.5.2.), which is a mixed crystal phase with the final link 2 CaO · Fe203, where the Fe can be continuously replaced by Al. As such, this line of mixed crystals is capable to exist also when exceeding the mole ratio Al203 : Fe203 = 2: 1. However, in Portland cement clinker, containing only compounds with the highest lime content, this line ends incidentally at a ratio of about 1 : 1; if the clinker contains more alumina, that part of alumina exceeding this ratio – which complies with the formula 4 CaO · Al203 · Fe203- is bound in the compound of the more lime containing tricalcium aluminate.
In spite of that tricalcium aluminate reacts very quickly with water, it does not show strong hydraulic properties, but in connection with silicates, it increases the initial strength of cement. The contribution to hydraulic hardening of the calcium alumino ferrite is minor [7b].
As was already mentioned in section 1.4.3., alka lies are bound by the clinker phases only if the amount of so3 present in the clinker, is not sufficient for complete binding in the form of alkali sulfate. They enter all clinker phases, but prefer the alumina phase. Also in this phase they are bound as mixed crystals, whereby the composition of the formula quoted in table 1.5.2. can be attained only when Si02 is simultaneously inserted [12b].
Potential clinker composition
The chemical analysis presents a picture of the com position of the oxides in the clinker or cement. R. H Bogue [13] developed a calculating method, according to which the content of the clinker minerals, (above all C3S, C2S, C3A, and C4AF) can be derived from the chemical analysis. It should be mentioned that the clinker composition calculated according to this method was denoted by Bogue as a potential composition. Potential means possible but not actual, it may then be said that the potential clinker composition determined by Bogue’s calculating method might possibly not be equal to the actual mineralogical composition.
Because of the clear presentation of the clinker com position and the possible prediction of cement properties, Bogue’s calculating method is in common usage. This method of calculation already appears in the cement standards of the United States, the USSR, and in those of many other countries. However, the ASTM cement standards of the USA contain a restriction saying that expressing of chemical limitations of calculated compounds does not necessarily mean that the oxides are actually or entirely present as such compounds [14].
then the following calculation can be performed: To begin with, we have to realize that c3s contains
73.69 % CaO, and 26.31 % Si02, and C2S contains 65.12% CaO, and 34.88% Si02. Table 1.6.1. also shows the composition of C3A and C4AF. Now we can set down the following proportions (see Table 1.6.1.):
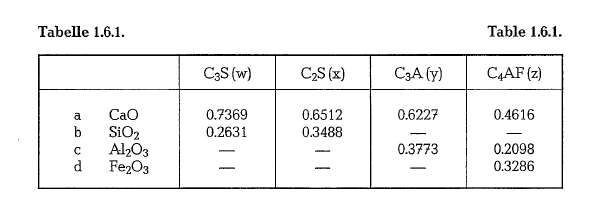
Hence, in any mixture of the four compounds, the CaO in the C3S is equal to 0.7369 times the per cent of C3S; the CaO in the C2S is equal to 0.6512 times the per cent of C2S, etc. The total CaO is then the sum of these values, or:


The compounds in other systems may be calculated by a similar procedure. In practice the following phase compositions can be encountered:

In the USSR there is another calculating method in use for the determination of the phase composition, namely by using Kind’s lime saturation formula

Investigating cement properties, Brown [17] determined microscopically the mineral content of various clinkers, simultaneously applying Bogue’s calculating method for comparison. The maximum values which Brown found using both methods of determination, are enumerated in table 1.6.2.
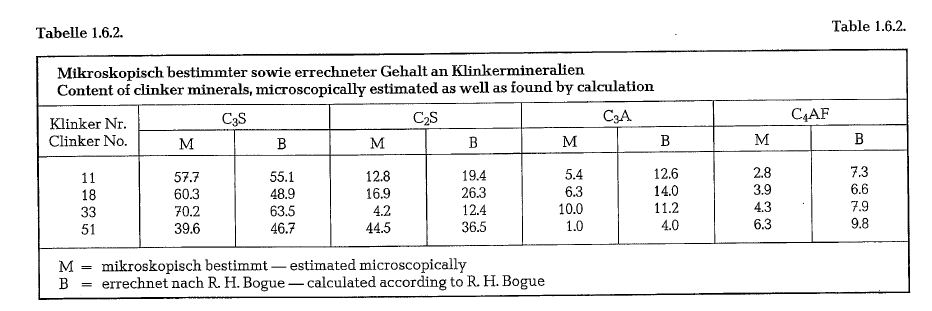
Nevertheless, practical experience justifies the statement that qualification of cements based on calculated clinker minerals is of sufficient usefulness.
In the USSR a chemical wet analysis method was developed for the direct quantitative determination of C3S, C2S and C3A. This method depends on the different solubility of these minerals in boric acid and in acetic acid [18].
Cement modules
For a long time Portland cement was manufactured on the basis of practical experience collected from the process of production. When comparing chemical analyses of Portland cement, it was found that certain relations exist between the percentages of lime on the one hand and the combination of silica, alumina and iron oxide on the other. Later, this oxide ratio gave rise to the formulation of the so-called
Hydraulic modulus
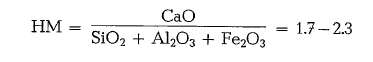
The hydraulic modulus of good quality cements was approximately 2. Cements with HM < 1.7 showed mostly insufficient strength; cements with HM = 2.4 and more had poor stability of volume.
As the formula demonstrates, the hydraulic modulus characterizes the cement by the ratio of CaO, to the total of the hydraulic factors, i. e. Si02, Al203, and Fe203• The HM is generally limited by the values 1.7 to 2.3. It was found that with an increasing HM, more heat is required for clinker burning; the strengths, especially the initial strengths step up and also the heat of hydration rises; and simultaneously the resistance to chemical attack decreases. At times the hydraulic modulus is still used. Later, for a better evaluation of the cement, the silica ratio and the alumina ratio were introduced; to a certain degree these ratios supplement the hydraulic modulus.
silica ratio
The silica ratio represents the proportion of Si02 to the total of Al203 and Fe203 :
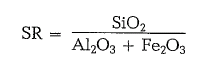
Generally, the silica ratio runs between 1.9 and 3.2. Preferred figures of the silica ratio are between 2.2 and 2.6. Now and then also higher figures for silica ratios can be met with as e.g. 3 to 5 and sometimes even beyond that, specifically for high siliceous cements and for white Portland cements. Likewise, low figures for silica ratios can be accepted as low as 2 and down to 1.5. An increasing silica ratio impairs the burnability of the clinker, by reducing liquid phase content and tendency toward formation of coating in the kiln. An increasing silica ratio also causes a slow setting and hardening of the cement. With a decreasing silica ratio the content of liquid phase increases; this improves the burnability of the clinker and the formation of coating in the kiln.
Silica acid ratio
![]()
When burning clinker in the rotary kiln, favorable coating conditions are created in the burning zone when the value of this quotient is limited to 2.5- 3.5, simultaneously keeping the value of the alumina ratio between 1.8-2.3. The silicic acid ratio should not be confused with the silica ratio discussed previously.
Alumina ratio
The alumina ratio characterizes the cement by the proportion of alumina to iron oxide:
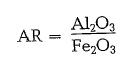
Generally, the values of the alumina ratio are in the range from 1.5 to 2.5. High alumina cements show an alumina ratio of 2.5 and more. The alumina ratio of low alumina cements is below 1.5 (so-called ferro cements). The alumina ratio determines the composi tion of liquid phase in the clinker. Where the alumina ratio is 0.637, both oxides are present in their molecu lar ratios, and therefore only tetracalcium alumino ferrite can be formed in the clinker (4 CaO · Ah03 · Fe203); consequently, the clinker cannot numerically contain tricalcium aluminate (3 CaO · Al203). This is the case with the so-called Ferrari-cement which is characterized by low heat of hydration, slow setting, and low shrinking. A high alumina ratio together with a low silica ratio results among other things, in a fast setting of the cement; this requires the addition of a higher gypsum rate to control the setting time.
Lime formulas
Lime saturation factor
To attain complete lime saturation in the clinker, the total silica must be combined as C3S, all iron oxide must combine with the equivalent amount of alumina to C4AF, and the remaining alumina must combine to C3A. Expressed in parts by weight:

The same result can be obtained if the C2S in the Bogue-formula equals zero, or, in the Kind formula (see below) KSK = 1.
Then the lime level in a clinker is characterized by the lime saturation factor (LSF), i.e. by the ratio of the effective lime content to the maximum possible lime content in the clinker.
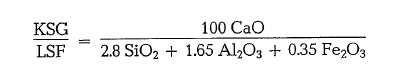
Kind’s formula, which is in use in the Soviet Union is based on the assumption that the incomplete lime saturation results only from a lower degree of binding between lime and silica
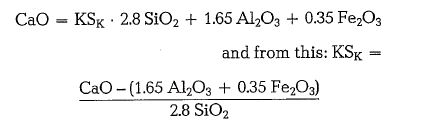
For technical purposes, the rate of the lime saturation factor, according to the above formula, fluctuates between 0.80 and 0.95.
In clinker showing a high iron oxide content (alumina ratio, AR < 0.64), alumina is bound only in the mixed crystal phase C2(A,F), and the maximum lime content as well as the lime saturation factor are:
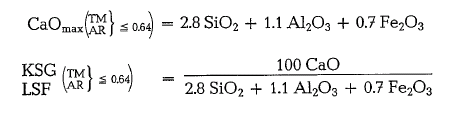
So also, the factors in Kind’s formula change if
AR< = 0.64.
Lime standard
The lime saturation discussed in section 1.8.1., as well as Bogue’s phase calculation shown under 1.6., are based on the assumption that from the sintering temperature the clinker cools down at such slow rate that during crystallization the liquid phase can achieve equilibrium with the solid phases.
However, this is not the case with clinkers containing C3A. At the sintering temperature of about 1450 oc, the silicate minerals C3S and C2S, and possibly not transposed free lime are in a solid state, whereas C3A and C4AF are in a state of fusion. However, the liquid phase is lower in lime than it would result from participation in C3A; to complete the C3A, the lime defi ciency can be restored by extracting the lacking lime during the crystallization process from the solid phases, which simultaneously are the most richest in lime, namely free lime and C3S [18a]. However, these processes cannot be completed during fast technical clinker cooling; practically, the liquid aluminate can not bind more lime than it already has absorbed atsintering temperature (“frozen equilibrium”, accord ing to Kiihl). Experimental investigations showed that the most lime saturated liquid aluminate binds prac tically two molecules CaO per Al203. Therefore, under technical conditions, this is the attainable lime limit, the so-called “standard lime”
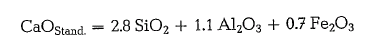
This formula shows ‘the same coefficients as the lime saturation factor (section 1.8.1.) with an alumina ratio AR 0.64. From this we get the lime standard as the ratio of the real lime content to the standard lime:
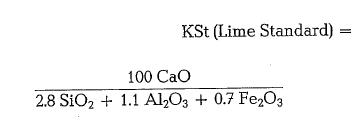
Later this standard was denoted as Lime Standard I (KSt I), to differentiate this formula from the more precise investigations based on the quaternary system CaO- Si02 – Al203- Fe203 and denoted as Lime Standard II (KSt II):
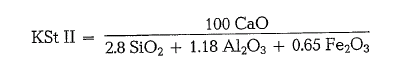
The different coefficients result from precise examinations which showed that in the liquid phase 2.15 molecules of CaO are bound per Al203 and that according to calculation, to binding the Fe203 remain only 4-2.15 = 1.85 molecules CaO.
Recently, a further refinement of Kiihl’s lime stan dard formula was proposed, taking into consideration the MgO-content [21].
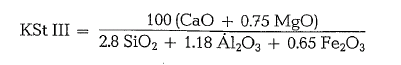
For the refined formula, MgO-contents only of up to 2 % can be used, since above this limit, the MgO appears as periclase (see 1.4.1.).
Generally, the KSt II is commonly in use [7b]. This value complies exactly with the British “Lime Satura tion Factor”. It has the same meaning as the German Lime Standard (KSt), but is not the same as the Ger man Lime Saturation Factor (KSG). For TM<=0.64, there is no difference between KSt and KSG.
The “Lime Saturation Factor” is also part of the Bri tish Standard Specification, and serves for the defini tion of the admissible lime content [23].
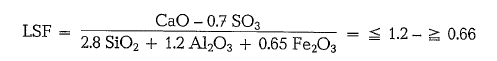
In this formula the LSF refers to the finished cement. The factor 0.7 S03 in the numerator means that the CaO-content which equals the analytically estimated S03-content, should be subtracted from the total CaO-content. In connection with this it is assumed that the total so3 comes from the added gypsum and not from the clinker.
A high lime standard normally causes high cement strengths. The following lime standard values are characteristic for Portland cements:
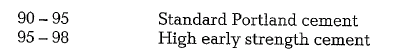
Free lime is caused by a lime standard above 100. However, a high lime standard requires a higher heat consumption for clinker burning.
Other modules
In France the lime content is rated according to the socalled hydraulic index (indice d’hydraulicite); it reads (in mols) [3]:
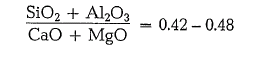
In the following more formulas may be quoted: The next one is an a,ttempt to express the known silica ratio in another way [3]:
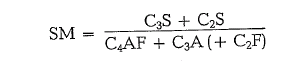
An increasing SM intensifies the resistance against chemical and atmospheric attack and causes princi pally higher cement strength.
Hardening ratio:
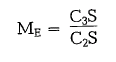
With an increasing ME there is an increase in the ini tial strengths of the cements, an increase in heat of hydration and a reduced resistance to chemical attack. The ME of standard cements is above 0.5. High early strength cements have an ME of approximately 8; cements with ME < 0.5 are belitic cements and tend to self disintegration of the clinker.
A combination of both moduls, i. e. Ms and ME shows a closer connection between these moduls and C3S and c2s respectively:
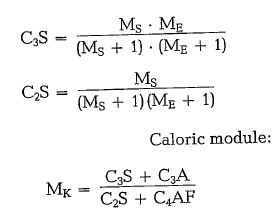
An increasing MK causes an increase in the heat of hydration of the cement. Quoted values for MK are within the limits of 0.3 and 1.8.
Thank you for your valuable information about raw materials. It is very useful for fresh specialists in the field of cement industry.
Very interesting can to my email below cause i wanna take deep reading if this article.
press CTR + P
this will make you able to save the article
thanks
how do we cite your article? thanks!
very useful information thanks
The literature is sufficient for beginners n also very useful for working professional in cement industry.
excellent….great work
Your e- book is excellent but I’d like to get also the size of additive raw materiels before grinding without limestone
Best regards,
Albert Kasongo
Very useful sir
hi
Please what is the MOST important material for making cement? Is it gypsum or limestone?
I need the clarification for my pupils . I would like to know their uses and percentages
Hi,
I would like to know how the percentage of raw materials that are needed to create for example white cement? I know that I can differ from the quality that the company wants to reach, but I just need an approximation. White cement consists of limestone which portions of 80%. Clay and silica sand 18% 12% other ingredients namely Gypsum and 3%.