Contents
click here to Download the Most Important 13 Books in Cement Industry
click here to Download the Most Important 13 Books in Cement Industry
How does magnesia bricks hydration occur? How does it affect negatively on bricks service life?
By: Nael Shabana qatar.cement@yahoo.com
Generally; magnesia bricks (magnesia-, magnesia-chromite-, and magnesia-spinel-bricks) must be stored in ventilated and dry rooms with protection against weather conditions (snow, rain, splashing water, etc.) as they are sensitive to hydration and can be damaged if stored improperly. In 2002 the commissioning of an ilmenite smelter on the North Coast of South Africa was extended by three months due to the failure and subsequent replacement of the magnesia-based refractory lining. The lining failed due to the hydration of magnesia caused by an unexpected source of water. The incident resulted in significant financial losses and a prolonged insurance claim which was settled in 2009.
Hydration of magnesia (MgO or periclase) in refractory material occurs when the material comes into contact with humid air, water, or steam. This exposure can occur during storage, construction, or operation. The optimal conditions for this hydration occur when water is present at 40°C to 120°C. This process is characterized by the transformation of magnesia into magnesia hydroxide according to this reaction: MgO + H2O à Mg(OH)2
The rate of the reaction depends on temperature, the magnesia content of the
brick, and pressure (if water is present in the vapor phase). The hydration rate with liquid water is slow, but as soon as the water penetrates as steam the
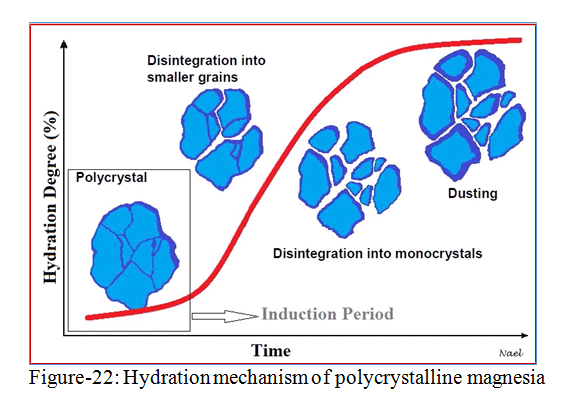
hydration becomes faster. Hydration of magnesia to magnesium hydroxide or brucite (consist of powder) results in an increase in volume of the bricks due to density change up to 115% (figure-20).

Figure-21 shows how extensive hydration leads to crack formation in the bricks and can subsequently lead to disintegration of the whole brick.

In industry, this mechanism is referred to as ‘dusting’ (figure-22). On the other hand, the volume expansion leads to brickwork movement which can affect directly on refractory lining inside the kiln. Hydrated bricks should not be used during construction and kiln shouldn’t be operated if such bricks are present in the structure.
Magnesia hydroxide decomposes thermally at approximately 330°C, and the last traces of water are expelled at higher temperatures to yield MgO as shown in the reaction: Mg(OH)2 >> MgO + H2O
Although the reaction is reversible, the damage to the bricks in the form of
cracks has already taken place. Reversing this reaction will therefore result in more porous bricks, which will lead to an increased and deeper molten material penetration.
The most important steps need to be considered to avoid or reduce the possibility of magnesia bricks hydration are:
1). Magnesia bricks must be transported in a container to protect the material against moisture.
2). Magnesia bricks should be stored inside storage rooms where it is dry, free of frost, ventilated, and with a temperature between 10°C and 30°C (see figure- 23). Note that magnesia bricks storage underneath a tarpaulin cover outside is not sufficient.
3). Bricks may not be stored for more than four weeks prior to installation and preheating.
4). Lining should be protected against moisture during installation and preheating.
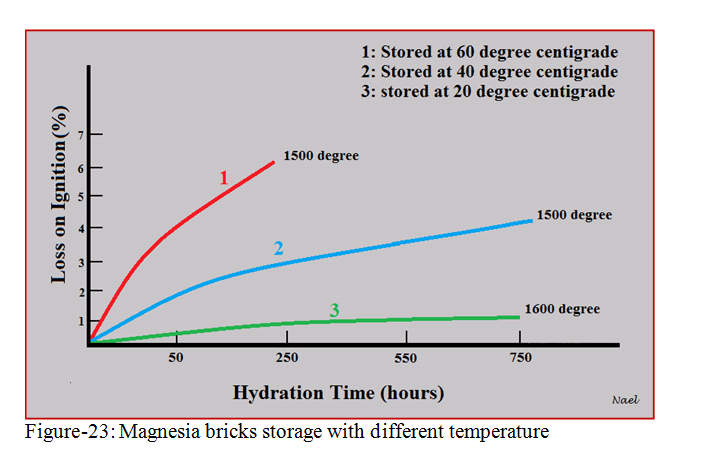
Figure-23 is showing the relation between magnesia bricks storage temperature and the degree of hydration in term of “loss on ignition”. As the temperature of storage room reduce, the hydration degree of magnesia bricks reduce.
click here to Download the Most Important 13 Books in Cement Industry
click here to Download the Most Important 13 Books in Cement Industry