Contents
click here to Download the Most Important 13 Books in Cement Industry
click here to Download the Most Important 13 Books in Cement Industry
What is the mechanism of kiln shell corrosion behind the refractory lining?
By: Nael Shabana qatar.cement@yahoo.com
Corrosion of the rotary kiln shell behind the refractory lining has become an increasingly serious problem for cement industry as it acts silently and reduces the shell thickness to below critical structural and mechanical limits of stability of kiln shell. Understanding the factors influencing the kiln shell corrosion and its formation mechanism is the best way to inhibit it. Corrosion of kiln shell behind the refractory is influenced by a number of factors such as composition of the metallic shell and its environment, temperature of the shell, cleanliness or roughness of the shell surface, its contact with other materials and severe process conditions. Carbon dioxide and sulphur dioxide are active scaling agents of iron and steel. The main reason of shell corrosion can be attributed to alternate oxidation at high temperature and acidic reaction at low temperatures when the kiln is stopped for repairs. The corrosion phenomenon takes place mainly due to presence of oxides, chlorides and sulphide at high temperature. There are three different kinds of kiln shell corrosion are observed in cement kiln systems:
1- Corrosion during kiln operation resulting in scaling of the kiln shell by oxidation of the metallic iron at elevated temperature (so-called high temperature corrosion) as shown in figure-31/A.
2- Rusting during longer kiln shut downs caused by condensation or absorption of humid (moist air, water) as shown in figure-31/B.
3- Wear of oxidized kiln shell during longer kiln shut downs caused by small blackflies eating iron compounds (called Ferrum Pedis syndrome) as shown in figure-31/C.

The reactions inside the kiln are different from reactions on the kiln shell surface since both the temperature and atmosphere are different. One of the most important reactions in the lining is the oxygen consumption where SO2 consumes oxygen and condenses as SO3:
2SO2 (g) + O2 = 2SO3 (↓)
The SO3 formed condenses as calcium or magnesium salts. The result can be that an oxidizing environment inside the kiln turns into a reducing environment at the kiln shell.
Figure-32 is showing the mechanism of kiln shell corrosion which passes mainly through four phases:
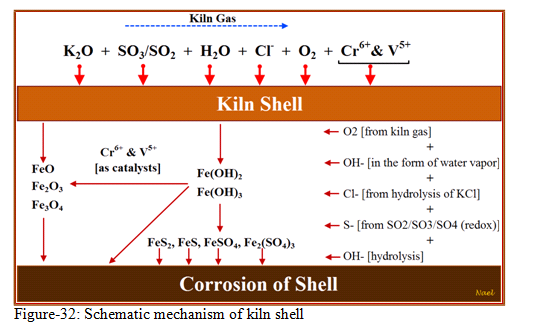
- Oxidation under high temperature:
In an oxidizing atmosphere, the iron from the steel shell will react with oxygen to form an oxide scale. Generally, this oxide scale is formed by more or less firm layers of different iron oxides. The compound with the highest oxygen content (Fe2O3) finds at the scale-brick interface and the compound with the highest iron content (FeO) at the metal-scale interface.
- Sulphidization under high temperature:
When no oxygen is present, SO2 takes over as the oxygen donor and a different reaction occurs. The reaction may be written as follows:
4Fe + 2SO2 (g) = Fe3O4 + FeS2
Accordingly, a sulphidization reaction can be identified by the occurrence of either pyrite (FeS2) or pyrrhotite (FeS) in the corrosion products.
As sulphide layers are more porous than oxide layers, the corrosion rate of the shell will increase. However, experience from different plants shows that, as long as chlorides are not present, the corrosion rate stays at an acceptably low level.
- Chloride attack under high temperature:
Sulphidization is enhanced by the presence of chlorides, mainly because they affect the morphology of the corrosion scale, hindering the formation of a strong, protective oxide layer.
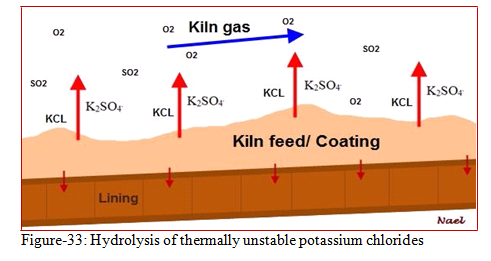
The total reaction is a chain process taking place at different temperatures. A temperature gradient between the kiln atmosphere and the kiln shell is created by the porous deposit and the refractory lining. The first reaction of the chain process takes place in the kiln and can be described as high temperature hydrolysis of the thermally unstable alkali chlorides (KCl) to form the more stable sulphates as showing in figure-33 and the reaction below:
2KCl(g)+H2O(g)+SO2(g)+½ O2(g) = K2SO4+2HCl(g) [T > 900oC]
This reaction step is followed by re-oxidation of hydrogen chloride gas (by oxygen or SO2) at lower temperatures to produce elemental chlorine (Cl2) which attacks the kiln shell.
2HCl (g) + ½ O2 (g) = Cl2 (g) + H2O [T < 400oC]
Once Cl2 (g) is formed, it can reach the kiln shell through the refractory bricks or through the gaps/joints within and between rings and will react with either the oxide-sulphide layers or (most likely) directly with the kiln shell according to the following reactions resulting in the corrosion of the kiln shell:
FeS+Fe3O4+4Cl2 = 4FeCl2+SO2+O2 [Reaction with the oxide-sulphide layers] Fe+Cl2 = FeCl2 [Reaction with the kiln shell]
4- Hygroscopic material attack:
During kiln shutdowns, deposit of salts containing potassium chloride in particular on the shell becomes active because being hygroscopic (absorbs atmospheric moisture). The chloride can reach the kiln shell in the form of gases but alkalis can only penetrate refractory lining as a part of liquid potassium and Sodium salt melts. So if product of corrosion contain substantial amount of potassium and sodium, the form of corrosion is termed as hot corrosion indicating that liquid phase take part in reaction.
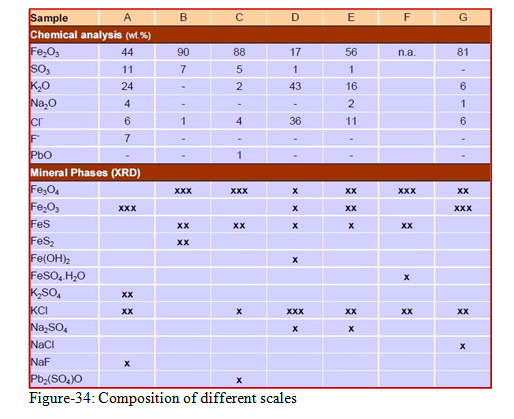
See the figure below (figure-34) which is showing the analysis of seven different samples of scales. Note the formation of Magnetite (Fe3O4), Hematite (Fe2O3), Pyrrhotite (FeS), and Pyrite (FeS2) in each scale sample.