Contents
click here to download Holcim , fls smidth , Lafarge , Most importnant manuals , most important excel sheets
click here to download Holcim , fls smidth , Lafarge , Most importnant manuals , most important excel sheets
Everything you need to know about combustion in Cement Kiln
Before entering into a detailed discussion on the combustion conditions in the kiln, it is appropriate to review some fundamental Jaws related to gases and combustion. Knowledge of these laws are essential in order for any kiln operator to understand the duties involved in his job. When malfunctions and unusual problems occur, such as chain fires, rapid and unusual temperature rises in tile fuel or firing system, an understanding of these Jaws combined with common sense can make the difference between a potentially serious, out-of-control condition and a safe solution.
GAS LAWS
All gases, including the hot gases in a kiln, behave in a certain manner under external influences. A perfect gas is one that obeys very closely cer tain physical laws. For all practical purposes, the gases under considera tion in this chapter may be assumed to be perfect gases. Adjustments to ti1e air circuitry of a rotary kiln are part of the responsibility of ti1e kiln operator, hence a basic knowledge of these Jaws is necessary to assist him in making the correct adjustments.
Before entering into a discussion of the gas laws, the reader is reminded that pressures and temperatures used in ti1e following equations are absolute. Absolute values are determined:
a) by adding 460° to the temperature in degrees Fahrenheit, g., 60°F = 520° absolute.
b) by adding atmospheric pressure to gauge pressure; while atmospheric pressure varies with altitude and weather conditons, it is sufficiently accurate for these computations to use 7, i.e., 50 psi gauge = 64.7 psi absolute.
In a like manner, CGS units are obtained by adding 273 :u the temperature in degrees centigrade, and 76 to the pressure in centimeters of mercury.
Boyle’s Law.
In 1662 Robert Boyle expressed tl1is law as follows:
Under constant temperature, tl1e volume of a given mass of gas varies inversely as the pressure upon it. In other words, pressure times volume, at constant temperature, is a constant expressed mathematically

Charles’ Law.
Similar to the discovery of Boyle, Jacques Charles in 1787 discovered that the volumes occupied by a given mass of gas at different temperatures are proportional to tl1e absolute temperatures of the gas provided the pressure remains constant.
All gases expand when the temperature is raised. If the temperature of a gas is raised 1 F, it will expand 1/460 of its original volume. In CGS units, a temperature rise of 1 C will result in an expansion of 1/273 of the original volume

Gay-Lussac’s Law.
Finally, there was Joseph Gay-Lussac who, early in the 19th Century, delved further into the action of gases, and developed the law that bears his name, which states that the pressure exerted by a given mass of gas will increase in proportion to the temperature if the volume is held constant. All unit again are in absolute values.

A General Law.
Now reviewing the above three basic gas laws, it becomes apparent that a general law can be stated, based on all three of the basic laws. The general equation is:
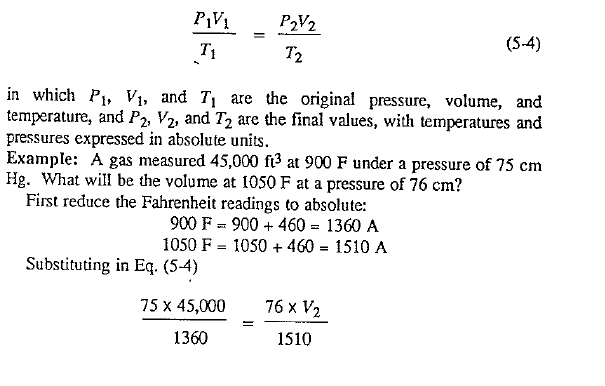
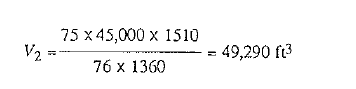
These gas laws can help a new operator to understand some of the fundamentals in kiln control. They are the solutions to some common basic control problems an operator might be confronted with on an almost daily basis.
THE COMBUSTION REACTION
Successful operation of a rotary kiln requires an adequate source of heat that will first raise the kiln to the desired operating temperature, and will then maintain this temperature by compensating for the various heat losses occurring in the kiln system, including the heat required for the process. The required heat is obtained by combustion of the fuel, a chemical reaction in which carbon, C, hydrogen, Hz, and sulfur, S, in the fuel combine with oxygen , in the air. To obtain combustion, two requirements must be fulfilled:
- sufficient oxygen must be present to mix with the fuel, and
- a certain temperature must be maintained to ignite the fuel oxygen
An operator must always remember the triangular relationship that leads to combustion:
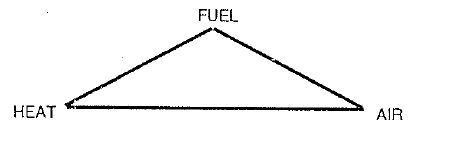
If any one of the three links is missing, no combustion will take place. If there is not enough air in the kiln, there will be no proper fire. Likewise in a dry or wet kiln, too much heat and air in the chain section will result in chain fire because chains contain carbon which acts as fuel. In order to stop a fire, the elimination of one of the components of the triangle is necessary, e.g., choking off the oxygen (air) supply. This will be discussed in greater detail later on. In addition, firing conditions inside the kiln must be such that the fuel particles undergo complete combustion while the fuel is still in suspension in the kiln atmosphere.
THE STANDARD COAL FACTOR, COMBUSTION AIR REQUIREMENTS
To determine the approximate combustion air needed to bum a given unit weight of coal, the formulas given below can be used when no ultimate coal analysis is available. The combustion air requirements here include 5 percent excess air.

Here it can be seen why gas- and coke-fired kilns tend to have their ignition point of the flame much further back in the kiln than coal or oil fired kilns. In addition, combustion in a kiln requires that sufficient time must be available to accomplish complete combustion while the fuel is in suspension in the kiln atmosphere.
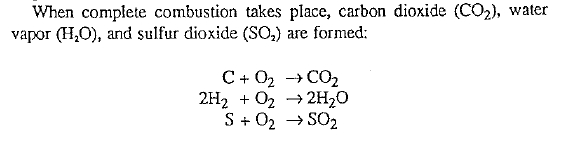
These equations for complete combustion indicate that when fuel is properly burned, 1 part of carbon will combine with 1 part of oxygen to form the combustion product carbon dioxide. However, when incomplete combustion takes place, then instead of carbon dioxide the combustion product is carbon monoxide:
![]()
Oxygen needed for combustion originates from the air which is forced into the kiln. Air consists mainly of 78 volumes (76% by weight) of nitrogen and 21 volumes (23% by weight) of oxygen. Thus, it is necessary to introduce approximately 5 volumes of air for each volume of oxygen needed to obtain complete combustion. The nitrogen contained in the air does not enter the combustion process, only the oxygen reacts with the carbon, hydrogen, and sulfur to form the combustion gases.
Effect of Kiln Air on Combustion Efficiency.
It should now be apparent to the readers that control of the air supply for the kiln is as important as control of the fuel rate, because each is dependent on the other in their effect on combustion. In the following it will be shown tl1at too little air (deficiency) as well .as too much air (excess) is harmful to the economical operation of a rotary kiln.
When combustion is incomplete because of a deficiency of air, approximately 4500 Btu are released when one pound of carbon is burned to carbon monoxide. However, under conditions of complete combustion in burning the same amount of carbon to carbon dioxide, 14,500 Btu will be released, a difference of I 0,000 Btu between complete and incomplete combustion, a clear indication of the importance of having at all times enough air available to accomplish complete combustion of the fuel. It should be pointed out that a rotary kiln, for safety reasons, is never allowed to be operated with such a deficiency in air supply. There is no·room for any compromise in such a situation; the operator must do everything in his power to again obtain complete combustion of the fuel within seconds after the first indication of incomplete combustion is given. It is definitely the wrong procedure to increase the fuel rate at a time when a deficiency of air is already present and the burning zone starts to lose temperature.
More common is a rotary kiln operating with a large percentage of excess air, usually resulting from inefficient attendance of the kiln by the operator. The question might be raised now why too much air is also harmful to the efficient operation of a kiln, the reasoning being that with plenty of air available a total burning of the fuel will with certainty be secured. However, operating a kiln with a large percentage of excess air increases heat losses to the rear of the kiln and lowers the flame temperature. Furthermore, any excess air not required for combustion consumes valuable heat because this air too will be raised to the operating temperature of the kiln. In short, by consuming heat to raise the temperature of the excess air, less heat will be available for the actual burning process of the material in the kiln.
G Martin,3 in an extensive study of the thermodynamics of rotary kilns, has calculated that for every 1% of free oxygen present in the kiln exit gases, there is a loss of 0.4449 tons of fuel for every 100 tons of coal burned. That is, 0.4% of the heat introduced into the kiln by the fuel will be lost for each I% free oxygen in the kiln exit gases. Consider, for example, a kiln producing 2040 tons clinker per day at an average specific consumption of 4.4 MBtu/ton clinker when the exit gas oxygen content is 1.5%. Burning the same amount of clinker per day but with 4.5% oxygen in the exit gas would result in a loss of 120 MBtu because of the excess air admitted to the kiln. At a common price of $1.80 per MBtu, roughly $67,000 per year would be lost
Determining Excess or Deficiency of Air.
The foregoing discussion naturally leads to the question: How does an operator know when the kiln is operating with too much or too little air?
To assist the operator, analyzers are available that continuously deter mine the amount of oxygen and combustibles (carbon monoxide) in the kiln exit gases. Other analyzers, less frequently used, are available for determining carbon dioxide content. The Orsatt apparatus is used for making periodical analyses of gas samples to determine oxygen, carbon monoxide, and carbon dioxide content
The percent oxygen contained in the kiln exit gases gives the best indication of the combustion condition in the kiln because this oxygen is directly related to the amount of air introduced and the amount of oxygen taken up during the process of combustion. We know that air contains 21% oxygen by volume. If there were no combustion reaction in the kiln, the same percentage of oxygen would be in the air leaving tl1e kiln. How ever, because combustion reactions do take place in the kiln, most of the oxygen reacts with carbon, hydrogen, and sulfur to form the combustion products CO, C02, H20 and S02. Thus, when no free oxygen can be found in the kiln exit gases no excess air has been introduced into the kiln.
The percent excess air can be calculated with the formula given by Perry, Chilton, and Kirkpatrick,4 provided that no combustibles (CO) are present in the kiln exit gases:
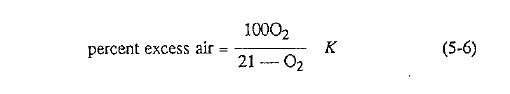
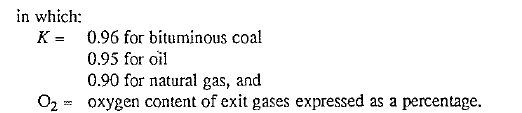
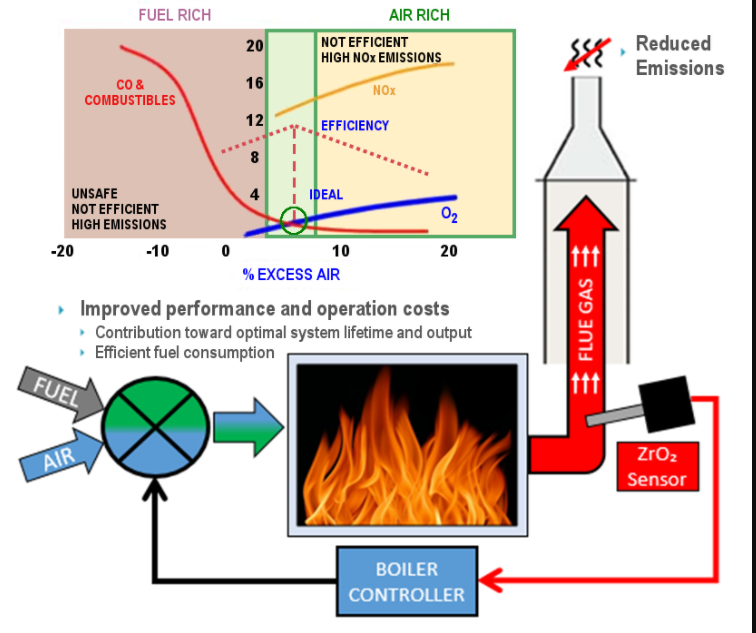
Fig. 5.1 shows this relationship for a typical rotary kiln.
The percent excess air can also be calculated from the results of a gas analysis obtained with the Orsatt apparatus. The following formula is especially helpful when a gas has a content of less than 1% oxygen because such gases usually contain traces of carbon monoxide also.
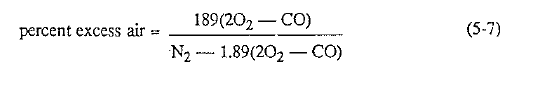
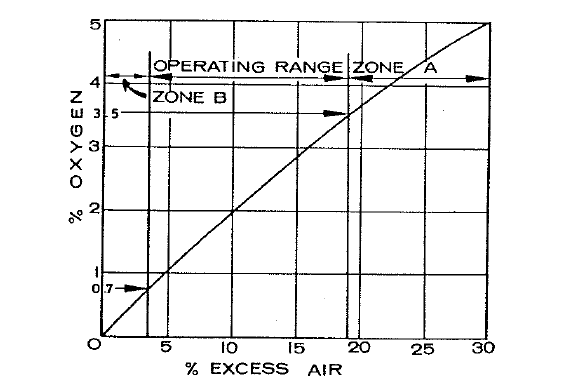
Fig. 5.1 Ideal operating conditions in the kiln occur when the kiln exit gas contains between 0.7 and 3.5% oxygen. Zone A indicates an excess of air, resulting in excessive heat loss; Zone B indicates a deficiency of air, resulting in the formation of carbon monoxide.
in which
02 = percentage of oxygen
CO = percentage of carbon monoxide, and
N2 = percentage of nitrogen
Incomplete combustion is easily recognized when any percentage, regardless of how small, is indicated on the combustible (CO) recorder. As stated earlier, a rotary kiln is never allowed to operate under conditions of incomplete combustion which means that at no time should there be any carbon monoxide present in the kiln exit gases.
Now consider the question of just what constitutes the ideal operating condition for efficient combustion and kiln control. At this poin theory and practice are apt to take divergent paths. Many viewpoints and opin ions, some good, some in error, have been dvanced, so it is well at this point to review the theory of perfect combustion. First, most efficient combustion takes place when there is neither carbon monoxide nor excess air present in the kiln exit gases: that is, the oxygen and the combustibles recorders should botlh have a reading of zero. Second, with any increase of
either carbon monoxide or excess air, valuable heat is lost.
Application of this tl1eory is of value only when perfect combustion conditionS prevail within the kiln burning zone. In practice, however, this condition is rarely attained because many factors associated with design of the kiln work against such ideal conditions.
A typical example that spotlights this fact is the common observation that a small percentage of carbon monoxide can be found in the exit gases while at the same time there is also a small percentage of free oxygen pres ent. This is in direct contradiction to the combustion theory that carbon monoxide shows only after all the excess air has been used up; that is, after the oxygen level has fallen to zero. In many instances the Orsatt analysis or the recorders have shown combustibles at oxygen· readings in the rang·e of 0.7%. This indicates that inefficient conditions are present when oxygen drops below 0.7%. The operator can observe the effects of changes in oxygen on tl1e flame itself. If, for example, the kiln is operat ing at an oxygen content of 0.7% and the fuel rate is increased without an increase in the air flow in the kiln, such an action will cause a change in the color of the flame, the flame taking on a darker color at its outer rim, a sure sign that t11e flame temperature is dropping.
Through experience it has been found that a rotary kiln operates best when the kiln exit gases have an oxygen content of not less than 0.7% and not more than 3.5% under stable operating conditions. The optimum target point is between 1.0 and 1.5% oxygen. In addition, under no circumstances should there be any carbon monoxide present in the kiln exit gases. The given targets and ranges for oxygen levels do not apply at times when the kiln is in an upset condition.
New and more advanced types of gas analyzers have recently been installed in many kilns. These advanced technology analyzers are capable of detecting and recording minute traces of gas components such as 02> C02, CO, as well as S02 and NO,. Recordings on these units are usually in terms of ppm (parts per million) instead of percentages.

The thing to remember is that conventional gas recordings will only indicate the presence of CO when it exceeds approx. 500 ppm whereas the newer analyzers are capable of recording contents as low as 10 ppm. It is therefore necessary to reword the above-mentioned operating rule and state that immediate counteracting moves must be made whenever the kiln gases contain more than 100 ppm (0.01%) CO.
The fact that both too little and too much air for combustion is detri mental for fuel economy, has led to the practice in some plants of main taining a constant fuel-to-air ratio at all times when the kiln is operating, even reaching the stage on some kilns in which controllers automatically adjust the total mass of air entering the kiln whenever a change in fuel rate is carried out. Although such an approach is fully justified from the viewpoint of combustion efficiency, it is not conducive to overall operating stability of the kiln.
Furnaces with less complex reactions than a rotary kiln can operate efficiently under the above principle. In the rotary kiln, however, there are many more factors that have a direct influence on the stability of the opera tion. For example, an important factor that is neglected when the prin ciple of a constant fuel-to-air ratio is used to control a kiln, is the heat profile through the entire length_ of the kiln. A rigid application of this burning technique causes the temperature proftle to change toward the back end of the kiln.
Table 5.1 shows the results of a burning technique in which the temperature profile has not been considered. Examples A and C follow the recommended practice of controlling the oxygen level within a range of 0.7- 3.5%, and at the same time giving full consideration to the back-end temperature. Example A shows how the fuel rate and the air rate into the kiln are proportionally changed with succeeding increases in the kiln feed rate. With each feed rate increase there must be a corresponding increase in fuel rate and air flow rate as well as a rise in back-end temperature to take care of the increased production. This, then, is the procedure for stable kiln operation. Failure to make all these required increases could lead the kiln into an upset. There is little doubt that such an approach as shown in Example A will meet the approval of experienced operators.
Examples of upset kiln conditions are shown in B and C in which the feed rate remains constant. Example B shows the technique of maintaining a constant fuel-to-air ratio; that is, the oxygen percentage remains constant. This results in uniform combustion but very serious fluctuations in burning zone and back-end temperatures. Changes in back-end temperature result in uneven feed preparation (drying, calcination) in turn creating cycling operating conditons. Because ti1e feed rate is constant, heat distri bution should be unchanged to maintain the same drying and calcining conditons.
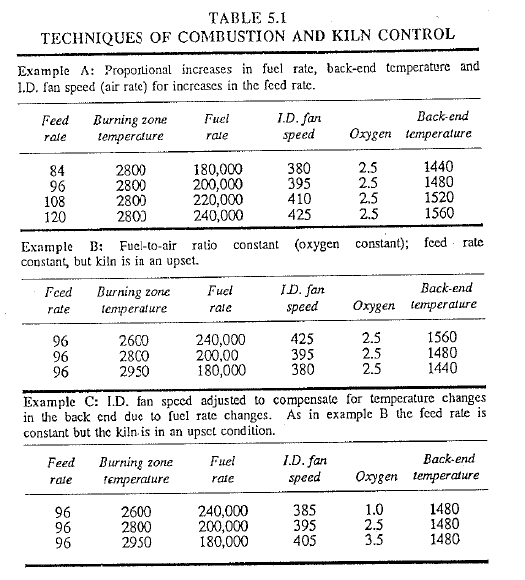
Example C is the recommended procedure for kiln upsets in which an attempt is made to maintain back-end temperature constant. Small adjustment are made to the induced draft fan speed to compensate for the temperature drops or rises that occur in the back end due to changes in the fuel rate.
The preceding discussion of combustion has centered on and is applicable to f1ring systems where the air supply to the furnace can be in dependently controlled. In other words, it applies to all kilns (we dry, semidry, preheater) that have one single-firing system in the lower part of the kiln (burning zone). It also applies to precalciner kilns that have ter tiary air canting from the cooler for the flash furnace. The discussion on the proper air supply to the kiln and the optimum percent of excess air does not apply to precalciner kilns where all the air for the precalcining chamber (flash furnace) originates from and goes through the rotary kiln (i.e., no tectiary air duct present). In such kilns one single air-supply route must serve two combustion processes namely the flames in the burning zone and the flash calciner. Obviously, the excess air at the rotary kiln back end must be much higher otherwise incomplete combustion will take place in the flash furnace. Excess air from 60-90% at the feed end is not uncommon on such kilns. Through experience it has become known that large percentages of excess air renders a kiln inefficient by lowering the thermal level of the gases. As a whole, such a precalciner might be efficient but the fact remains that the burning of the fuel in the burning zone is done by large amounts of excess air hence no optimum conditions prevail here. This, too, is the reason why a precalciner, without tertiary air duct, usually does not achieve the high output rates that a pr calciner of equal size with tertiary air duct does.
Combustion air requirements to bum fuel (solid or liquid) can be calculated as follows:
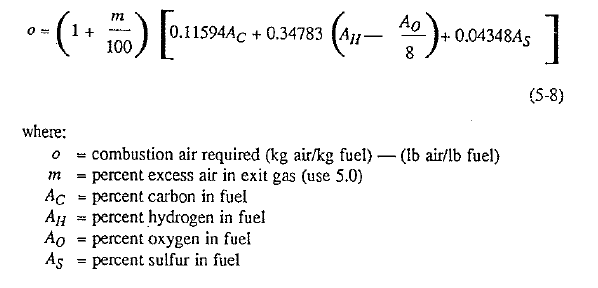
Products of combustion are mathematically detemined by the following formulas:

Carbon Dioxide in the Kiln Exit Gases.
The percent of oxygen in kiln exit gases is solely a function of the amount of air introduced into the kiln and the amount of air consumed in combustion of the fuel. In contrast to this, the percent of carbon dioxide in the exit gases is influenced by calcination of the raw feed in the kiln as well as by combustion.
When burning pulverized coal under perfect conditions, that is, when no excess air is present in the kiln gases, a maximum of about 18.3% C02 would result from the combustion if there were no evolution of C02 from the raw feed in the kiln. For fuel oil this value is about 15.8%, and for natural gas, 12.2%. These percentages are obtainable only when perfect combustion conditions prevail in the kiln, which is very seldom the case. Any excess air present will cause a reduction in the percent of C02 in the gases. However, analysis of kiln exit gases in a cement plant shows a percentage between 22 and 28% C02. Therefore, a portion of the C02 in the kiln exit gases must originate from calcination of the raw feed.
Assunting that the C02 originating from the kiln feed remains constant, then any drop in C02 in the exit gases indicates an increase in air in the kiln. Knowing that a rise in excess air results in poorer fuel efficiency, it is then true that a decrease in C02 in the exit gases indicates a higher heat consumption for each ton of clinker burned. Hence the operator should try to obtain the maximum percentage of C02 possible during normal operation of a kiln.
If the only function of the C02 analyzer were to indicate fuel efficiency, there would be no reason for its use as this information is available from the oxygen analyzer. The conditions under which a carbon dioxide recorder becomes of value is when calcining conditions change in the kiln, that is, when the kiln is in an upset state. A change in the feed rate (including the dust return rate, if any), feed composition, or feed advancement within the kiln, can result in a change in the C02 content in the exit gases even at constant combustion conditions in the burning zone. The reason why the C02 changes in these instances is because more feed, less feed, or different feed, is being calcined in the kiln.
Changes that occur in- the calcining zone cannot be seen by the kiln operator when he looks into the kiln to observe the burning zone because calcining takes place behind the burning zone. For this reason, a change in the character of the feed bed could occur unknown to the operator until it later becomes visible in the burning zone. Most common of such occurrences are kiln feed dust waves that flush at high speed into the burning zone, the arrival of large chunks of scale, or a change in the amount of feed entering the burning zone. By the use of a carbon dioxide analyzer and recorder, the operator is able to recognize such changes before they become visible in the burning zone, thus giving him time to make the necessary adjustments in the kiln control variables sooner than he could without the analyzer. If the operator notices an increase or decrease in the carbon dioxide recording, no adjustments having been made to such variables as fuel rate, air flow rate, or kiln speed, he knows that some change has taken place in the calcining zone. This is an early warning of possible changes in burnability that will soon occur. Once an operator has become accustomed to a particular kiln, he will know what settings in C02>02, fuel rate, feed rate, and other variables will most likely result in an efficient and stable kiln operation for a given feed composition ..
A carbon dioxide analyzer is also helpful in obtaining stable operating conditions again after a kiln upset. Assuming that the kiln has been in an upset condition with oxygen lower and C02 higher than normal, then when the recorders start to show an increase in oxygen, with the corresponding decrease in C02 without any change in feed rate, the operator will know that calcining conditions behind the burning zone are improving and he can expect an increase in burning zone temperature within a few minutes.
Proper combustion requires that the kiln be operated in such a manner that:
- The kiln exit gases have an oxygen content of not less than 7% nor more than 3.5% under normal operating conditions.
- The kiln exit gases contain no carbon
- The kiln exit gases contain the maximum percentage of carbon
In addition, the operator should:
4- Strive for optimum combustion conditions at all times by stabilizing kiln gas oxygen content between 0 and 1.5%.
5- Take immediate steps to eliminate combustibles in the exit gases whenever any carbon monoxide is detected therein.
In preheater and precalciner kilns it is of interest to note by how much (percent) the kiln feed is calcined after it leaves the preheater vessels and enters the rotary kiln proper. The laboratory checks this by analyzing the feed before and after the preheater tower, then calculates the result as follows:
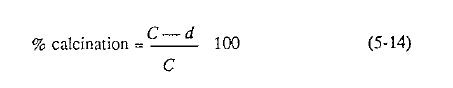

In the second group are the variables over which the kiln operator has control in order to obtain desired flame characteristics. These are:
Group 2 Fineness of the coal burned Temperature of the fuel oil Temperature of primary air
Temperature of secondary air
Flow rate of primary air Flow rate of secondary air Fuel rate
Burning zone wall temperature
Position of the burner
Degree of purity (dust concentration) of combustion air entering the kiln
Cross-sectional loading of the kiln
Air requirement of the fuel
Density of the primary stream (primary air plus fuel)
Density of the combustion gases
The nine italicized variables are the ones indicated by Gygi* as being the most important factors affecting the flame in a rotary kiln. A change in flame characteristics will take place whenever one of the variables in either group is changed. If the change results in a dangerous or undesirable flame, one or more of the other variables must be adjusted to counteract the bad flame condition.
Flame characteristics can vary considerably from one cement plant to another, sometimes even from one kiln to another in the same plant. The reason for this is that a flame must always be tailored to existing kiln designs and prevailing operating conditions. Clinker quality, refractory, presence of rings, and kiln equipment problems force an operator to obtain a certain flame that best fits the actual conditions in the particular kiln under consideration.
As a general rule in flame control, one should attempt to obtain the shortest possible fire and the highest possible flame temperature without adversely influencing clinker quality, coating formation, ring formation, and refractory life, or causing damage to the kiln equipment in the discharge area. Furthermore, the flame must not cause overheating in the burner hood, kiln discharge end, or cooler. Once the ideal flame characteristics have been obtained, the operator should make every effort to operate the kiln in such a fashion as to cause a minimum of disturbance to the flame. A flame should not willfully be changed during the course of kiln operation unless specific conditions necessitate a change.
Now consider in detail the several flame characteristics that were mentioned at the beginning of this chapter, and how these characteristics can be modified.
Please share me waste generator procwss.