Contents
click here to Download the Most Important 13 Books in Cement Industry
click here to Download the Most Important 13 Books in Cement Industry
EVERYTHING YOU NEED TO KNOW ABOUT CEMENT CHEMISTRY FROM ANCIENT TIMES TO 2019
Cements:- used since historical times (Pyramids » 3000 BC; Colosseum). Ancient cements of varying types. Two main classes of constructional cements are defined: non-hydraulic cements – do not set under water (see box), and hydraulic cement.

Non-hydraulic cements
were amongst the most common of the ancient cements. The relatively high solubilities of portlandite (Ca(OH)2) and gypsum means that they deteriorate rapidly in moist or wet environments. The early Romans made good use of lime based cements and mortars (cement + sand) by ramming the wet pastes to form a high density surface layer which carbonates in contact with air to produce a low permeability surface skin of calcite. This protected the underlying Ca(OH)2 and examples of Roman lime mortars can still be seen in Hadrians Wall. Lime mortars were still used in domestic construction until relatively recently. Raw (natural) materials required temperature treatment. Partial dehydration of natural gypsum (»200oC), and calcination of calcite (»850oC). Hydraulic cements – more durable. Hydration products are very insoluble – cements set under water. Earliest systematic development of these, probably Roman – use of limestones containing silica and alumina and also use of volcanic earths as an additive to limestone prior to calcination. Forerunner of modern Portland cements.
Portland cement –
patented by Joseph Aspdin in mid-1800’s. Made from finely ground limestone and finely divided clay to give a burned product containing 65-70% CaO, 18-24% SiO2, 3-8% Fe2O3, 3-8% Al2O3 plus smaller proportions of minor oxides (e.g. Na2O, K2O, MgO, etc.). Modern plants permit much more efficient processing and in addition, proportion raw mix compositions to produce a cement from which a range of strength development and durability properties can be expected.
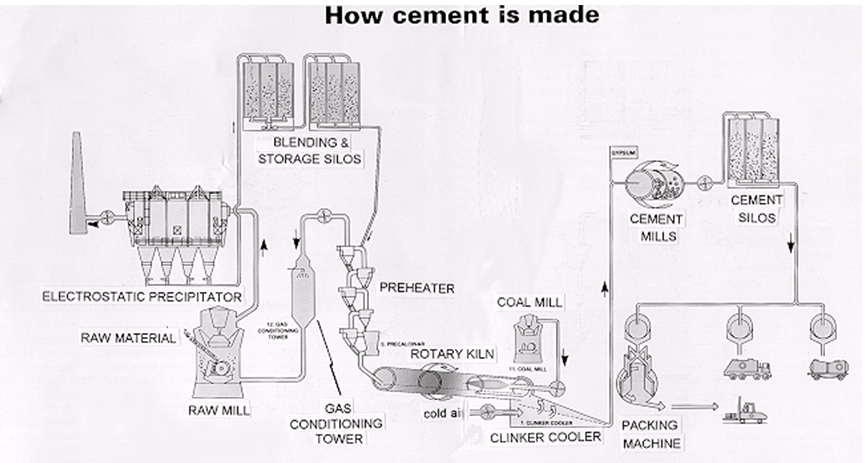
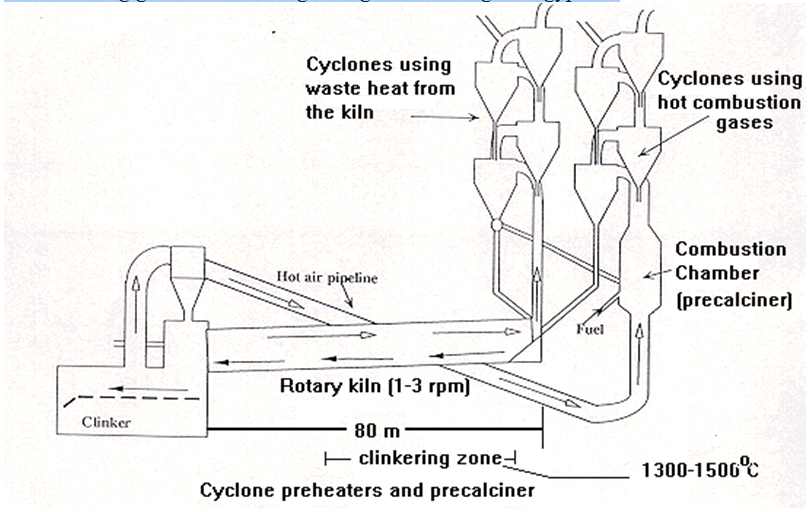
Efficient grinding and blending of raw materials is essential. Firing of blends (raw meal) takes place in a rotary kiln following drying and dehydration in cyclone preheaters and pre-calciners. Materials undergo solid state reaction and partial melting (clinkering) at temperatures of up to 1500oC (in the hot zone of the kiln). Cement clinker is recovered on the cooling grate and sent for grinding and blending with gypsum.

Cement clinker composition:
– main clinker minerals are alite, belite, aluminate and ferrite. Proportions vary depending on raw meal composition and firing and contribute to defining the hydration and strength development properties of the cement product.
Alite– C3S, 3CaO.SiO2 (idealised): Minor – Al2O3, MgO, P2O5, Fe2O3, Na2O, K2O
Belite– C2S, 2CaO.SiO2 (idealised): Minor – Al2O3, MgO, P2O5, Fe2O3, Na2O, K2O
Aluminate – C3A, 3CaO.Al2O3 (idealised): Minor – Fe2O3, SiO2, MgO, Na2O, K2O
Ferrite – C4AF, 4CaO.Al2O3.Fe2O3 (idealised): Minor – SiO2, MgO, TiO2, Mn2O3.
Chemistry of clinker formation:
– information on raw meal composition and required kiln conditions may be obtained from equilibrium phase diagrams. Minor components have a small influence on equilibria but approximations can be made using the CaO-Al2O3-SiO2-Fe2O3 diagram although it is perhaps preferable to restrict initial considerations to phase relations in the CaO-SiO2 and the CaO-Al2O3-SiO2 systems.
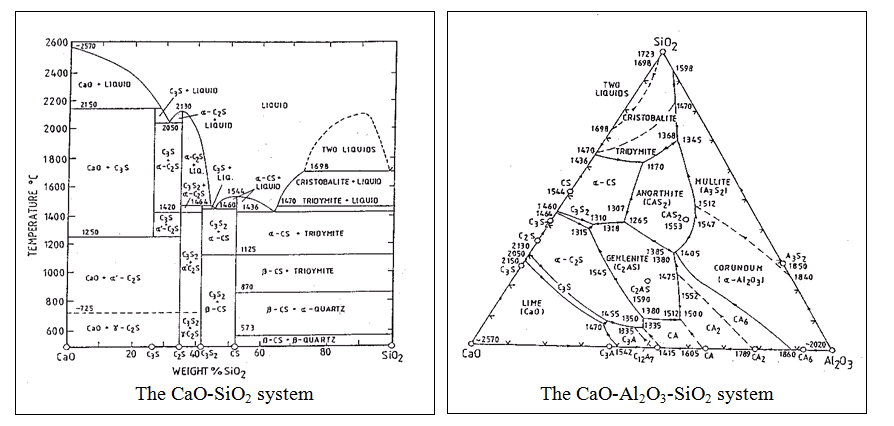
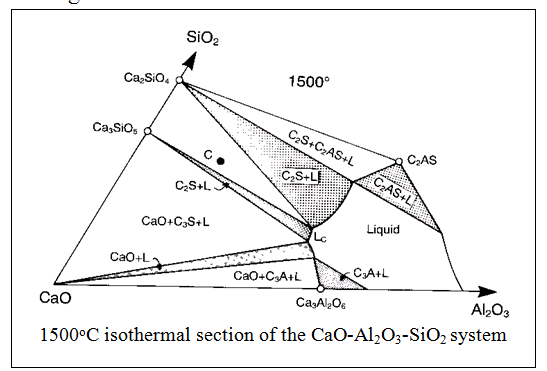
The CaO-SiO2 and CaO-Al2O3-SiO2 systems are binary and ternary systems respectively. The ternary system shows the temperature and compositional information of the binary system (CaO-SiO2) but with the third component (Al2O3) also, viewed from above. The curved lines represent temperature ‘valleys’ as on a map. Compositional information at a fixed temperature can be derived from an isothermal section. The 1500oC section of the CaO-Al2O3-SiO2 system allows some consideration of phase equilibria in cement making.
Approximate bulk composition of raw meal is represented by C which means that C2S and C3S will be in equilibrium with a liquid phase of composition Lc (which contains CaO, Al2O3 and SiO2). It would expected from this diagram that the liquid would cool to crystallise C3A. In practice, the final phase assemblage includes C12A7, a phase relevant to a lower temperature diagram. This arises due to the non-equilibrium effect of fractionation; as cooling occurs, slow reactions can cause high temperature assemblages to be frozen in). The final properties of the cement strongly depend on its mineral composition so that raw meal composition and firing conditions are adjusted depending on the type of cement to be produced (see later notes on Cement Type). The cement manufacturer expresses the product composition both as an oxide analyses (chemical) or mineral composition. The latter is calculated using the Bogue calculation.
HYDRATION
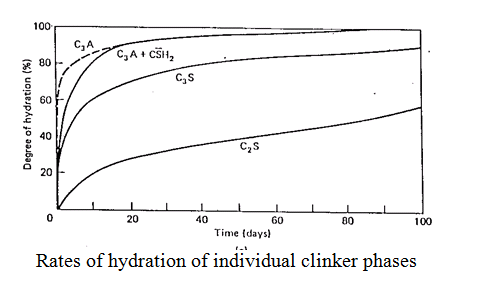
– the term used to describe a range of reactions between cement and water to produce a hardened product. A cement clinker particle is a multiphase solid having massive calcium silicate grains (50 – 100 mm) in a matrix of interstitial aluminate and ferrite. They each have specific reactions with water (see box) to produce a range of hydration products which intermesh and interleave to produce a dense and strength developing solid. The rates of reaction are important. The C3A reaction is fastest and also generates most heat (cement hydration is exothermic) but little contribution to ultimate strength is derived from this phase alone although it contributes significantly to early strength. The principal contributers to longer term strength are the calcium silicates. C3S is most reactive, giving early strength but C2S has a better longer term contribution. The C-S-H produced is the principal binding phase in Portland cements and is quantitatively the most significant hydration product. The ferrite reactions are intermediate in rate between the C3S and C2S reactions but have an important long term contribution to strength and durability.

Properties of Cement Hydration Products
Ca(OH)2 –
crystalline, isostructural with the natural mineral Portlandite. Solubility at 25oC of around 1g.l-1.
C-S-H
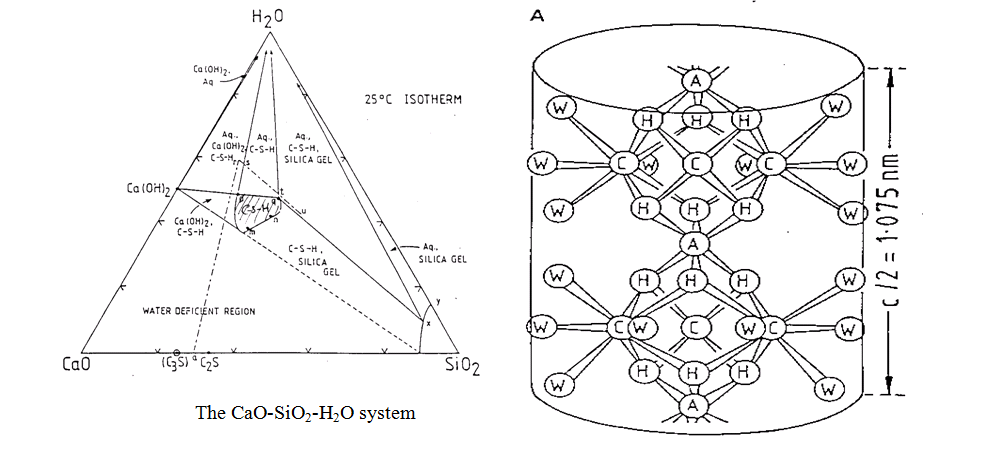
– poorly crystalline product of variable composition. Considered to be based on a crumpled layer structure (analogous to a distorted clay sequence) which traps regions of porosity – pore size distribution from nm to mm. Simplified composition given by:
CaxH6-2xSi2O7.zCa(OH)2.nH2O where CaO/SiO2 = (x+z)/2 (Glasser et al, J.Am. Ceram. Soc., 70, 481-5, (1987)). Variable CaO/SiO2 ratio (approx. 0.8 to 1.8) and variable H2O:SiO2 ratio (see CaO-SiO2-H2O phase diagram below). Variable composition means variable solubility properties. Solution compositions above C-S-H and the presence of other phases defines the C-S-H composition (see lectures) so that cements containing siliceous blending agents will have compositionally quite different C-S-H to that found in OPC pastes.
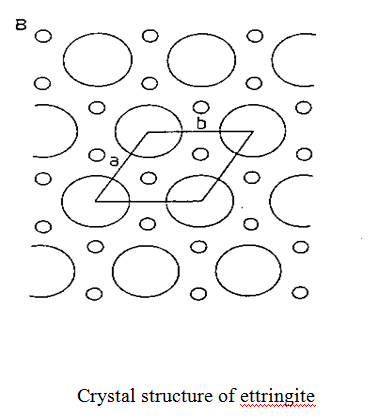
AFt
– or ettringite, or aluminoferrite trisulphate (C6AS3H32). Crystalline – trigonal. Forms columnar type structure consisting of (Al,Fe)(OH)6 octahedra alternating with triangular groups of edge sharing CaO8 polyhedra with which they share OH– ions. Inter-column regions contain loosely bound SO42- groups which are exchangable. Responsible for retardation of C3A hydration (due to coating of C3A).
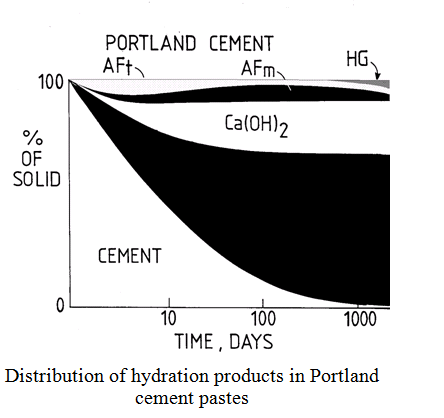
AFm
– or monosulphate. Crystalline – layer structure derived from that of Ca(OH)2 by the ordered replacement of one Ca2+ ion in three with Al3+ or Fe3+. These layers alternate with ones containing anions which balance the charge (e.g. SO42-, OH–, etc.) and H2O. Composition is [Ca2(Al,Fe)(OH)6].X.xH2O, where X represents the interlayer anion.
Hydrogarnet
– nominally C3AH6, but in practice contains Fe and Si. Related to the mineral grossular or garnet (Ca3Al2Si3O12) which has a cubic structure. Not normally a product of modern OPC hydration although present in blended cements and older Portland cements.
Aqueous phase and pore structure
– pore fluid permeates the microstructure of the hardened cement paste via the pore system. It is highly alkaline (pH > 13) due to rapid and almost quantitative dissolution of Na and K salts from the cement clinker. The porosity of the paste comprises of interconnected and isolated pores, the pore sizes of which are important to the strength and dimensional stability of cement products.
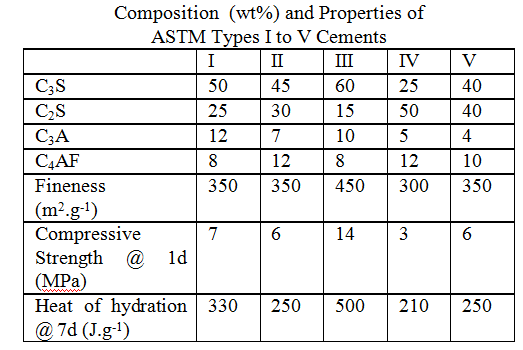
Cement Types and Standards:
provides some quality restrictions to cement compositions and performance. Different types of cement are used to meet different performance criteria. Properties can be estimated from compositions and fineness. Try to estimate which cement types will be rapid hardening, low heat ouput or sulphate resistant Portland cements.
Blended Cements
The use of cements in concrete which have not been blended with some form of reactive additive will become less likely in the future. There are now a range of additives commonly used to enhance the properties of concretes and, in some cases, result in reduced materials costs. This is mainly because they are industrial by-products. The most common of these are: pulverised fly ash (PFA)- a coal combustion product; blastfurnace slag (BFS), from iron making, and; condensed silica fume (CSF) from the ferrosilicon industry. The suitability of such materials depends on: their reactivity, their cost (availability) and their influence on the properties of the resulting concrete. All influence the internal chemistry of the cement system, i.e. pH, mineral balances, and their generally slower reaction leads to a longer equilibration time as reaction products of the additive re-equilibrate with cement hydration products. Reactivity, in all cases, depends on glass content, particle size, composition (nature of impurities) and external influences such as temperature, humidity and hydrating liquid composition.
Silica fume: quartz reduced in an electric arc furnace – some SiO volatilisation and oxidation produces largely glassy SiO2 particles of »100 nm diameter. Low density material with 86-95% reactive SiO2.
PFA: arises as a dust in chimney stacks above coal-burning power station furnaces. Have widely variable compositions depending on furnace operating conditions and coal source. Particle sizes may be low, with 50% < 10 mm or coarse with 50% < 40 mm. Particles are generally spherical (formed by rapid cooling from a melt) and may be hollow (cenospheres) with or without spheres inside (plerospheres). Largely glassy (85 – 90%) with small crystallites of mullite (A3S2) and quartz.
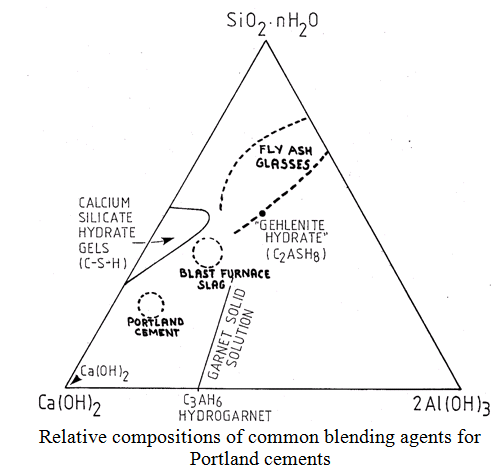
BFS: produced in the iron blastfurnace. Fluxes impurity oxides and sulphides. Composition is monitored (defines iron quality). Best for iron maker when S(CaO+MgO)/S(SiO2+Al2O3) is maximised. Mainly glassy (>95%) and has its own CaO content (approx. 40%).
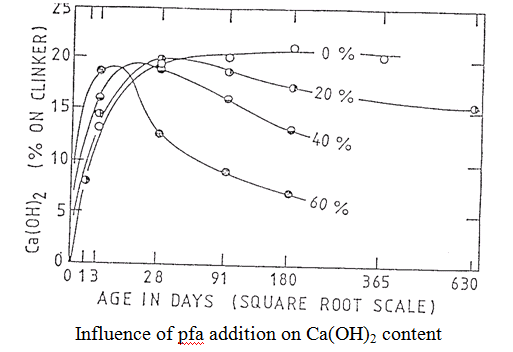
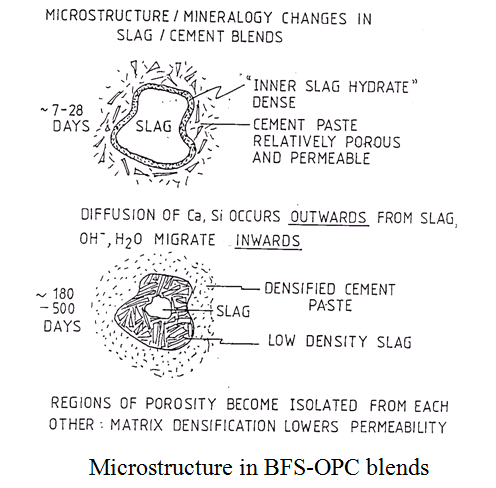
Hydration characteristics Silica fume reacts relatively fast in the cement system. Pastes require a higher water content than silica fume-free ones unless a superplasticiser is added. The silica is consumed in reaction with Ca(OH)2 and lime-rich C-S-H resulting in a paste with lower (or no) Ca(OH)2 and a C-S-H of low CaO:SiO2 ratio (maybe as low as 1.2). The nature of the CSF-cement reaction process leads to efficient pore-filling and consequent enhancement of mechanical performance (low porosity pastes are stronger than high porosity ones). Silica fume-OPC blends are therefore used in the production of High Strength Concrete (HSC) with compressive strengths routinely in excess of 100 MPa. PFA displays the same pozzolanic action as CSF (see plot above) but is generally much slower in reactivity due to the coarser particle size. Also, the alumina (around 30%) and iron (around 10%) content contribute to the formation of products other than C-S-H but even so, general C/S ratios can be significantly lower in PFA-OPC blends than in neat OPC. (Note that in all blended cements, compositional gradients are common.) Other products include hydrogarnet, e.g. C12A3FS4H16 was reported within a PFA cenosphere found in an aged paste (Rogers and Groves, Adv. Cements Research, 1, 841, (1988)). BFS has different hydration characteristics to CSF and PFA. Having a CaO content of its own, it is not a pozzolan as such (i.e. it does not depend on external sources of CaO to activate it – although its reactivity is accelerated by activators such as Ca(OH)2, Na2CO3, NaOH, etc). Typically, BFS exhibits an initial burst of activity in OPC-BFS blends. This is followed by a relatively dormant period which may last six months or more, depending on temperature, particle size and aqueous phase composition, before continued hydration consumes remaining glassy grains. Hydrotalcite (a magnesium aluminate hydroxide hydrate) and gehlenite hydrate (C2ASH8) are commonly found in BFS-OPC pastes along with ettringite, monosulphate and C-S-H but again, Ca(OH)2 contents are substantially reduced and C-S-H has lower Ca/Si ratios with respect to neat OPC. A unique characteristic of BFS is its electrochemically reducing characteristics. Eh values of < -500 mV vs SHE (standard hydrogen electrode) have been measured in pore fluids extracted from blends of 85% BFS – 15% OPC cured for 28 days. This compares with around +100 mV measured for equivalent OPC pastes. This feature has important implications for the use of BFS blends in waste immobilisation and for passivation mechanisms for steel.
Effects of Blending Agents on Paste Microstructures.
The durability of cement pastes is strongly influenced by: (i) internal chemistry, and (ii) paste microstructure. The industrial by-product additives above all influence the development of paste microstructures. In neat OPC pastes, two types of porosity contribute to the total pore volume. Isolated pores are completely enclosed by hydration products so that material transport into and out of the pore is limited. Connected porosity is that through which a continuous pathway between regions of the microstructure exists. Continuous or interconnected porosity often (although not always) links the interior of the paste to the outside world so that aggressive chemical species can penetrate and degrade the paste internally having consequences for paste durability. The effect of the blending agents identified above on microstructure is to cause a reduction in the degree of interconnected porosity. This is especially true in the case of BFS-containing pastes. Although the overall porosity, as determined by neutron scatterring is still significant, the interconnected porosity as measured by intrusion methods (e.g. MIP) is low.
DURABILITY – the ability of the product to resist changes imposed by its service environment. Durability includes influences of mechanical damage, e.g. abrasion, thermal expansion, but is more commonly associated with chemical effects, e.g. sulphate attack, chloride-induced or general corrosion of steel reinforcement, alkali-aggregate reaction, etc.
Sulphate attack: – expansion arising from the reaction between monosulphate (4CaO.Al2O3.SO3.12H2O) and SO42- in the presence of aqueous Ca2+ to give ettringite. The conversion from the high density phase to the low density one can cause expansion and cracking. The cracking opens up new connected porosity which accelerates the transport of sulphate into the cement paste and the deterioration of the paste. (Try writing out the equations for the monosulphate-ettringite conversion).
Delayed Ettringite Formation: – a fairly recently discovered problem relating to degradation in steam-cured products. A number of mechanisms for this have been proposed (and at least one major legal conflict has arisen based on assigning liability for failed concrete products). It is proposed that the temperature cycle used in steam-curing produces a non-crystalline ettringite precursor which, on cooling and after some extended time period, crystallises having absorbed the required amount of moisture.
Carbonation: – lowering of matrix pH due to the reaction between dissolved CO2 and the calcerous phases in the paste. Found at surfaces, a zone of carbonated product penetrates towards the interior to a distance which is defined by the porosity of the paste. Carbonation can ultimately consume Ca(OH)2 and C-S-H, the degradation of C-S-H leading to a progressive decalcification to very low Ca/Si ratios and ultimately, silica gel. Degradation of the principal binding phase therefore can lead to strength loss. A more immediate concern is the loss of high pH in the vicinity of steel reinforcement. A pH of greater than about 10.5 is thought to preserve a passive film on the steel, protecting it from corrosion. Loss of pH therefore increases the corrosion risk. Engineers specify a minimum ‘cover depth’ to attempt to deal with this problem but again, the depth of penetration is porosity dependent.
Effect of Chloride: – used to be a common additive to cements to accelerate setting but this use is now banned in structural concretes (Why do you think CaCl2 would accelerated cement setting?). Chloride interactions with set concrete are however common, e.g. de-icing salts, salt spray, etc. Penetration is via connected porosity so that cover concrete may quickly become heavily loaded with soluble chloride. Steel passivation films are rapidly de-stabilised by chloride locally giving rise to pitting corrosion and rapid deterioration of the steel (the corrosion is focused in series of a small areas). There are competing influences of alkalinity and chloride effects so that a useful parameter to monitor is the [Cl]/[OH] ratio.
Steel Reinforcement Corrosion: The oxidation of Fe(m) to Fe2+. In addition, further oxidation and cathodic reactions lead to production of oxides and oxyhydroxides of Fe (III) which produces a low permeability ‘passive’ film which slows the corrosion rate down considerably. Where corrosion can continue (by depassivation), expansion of corrosion products at the cement-steel interface and the subsequent spalling of cover concrete can occur. Many examples of this can be seen in concrete structures. Spalling leads to exposure of previously internal concrete as a fresh site for environmental damage.
Alkali -Aggregate Reaction (AAR): Certain rocks contain silica in a mildly reactive form. Generally, flints, opals, cherts and strained quartz have a high degree of reactivity in concretes. The reaction is driven by the high pH pore fluid and the reactive silica and gives rise to a sodium silicate gel product which contains only a small amount of calcium. The gel imbibes water causing swelling and this gives rise to expansion cracks in affected concretes. The degree of expansion is important with respect to the servicability of affected structures. A recent study on a water inlet tower of a dam in Tasmania, Australia, showed that AAR expansion had increased the diameter of the tower sufficiently so that the inlet valves were unable to stem the flow of water when they were in the closed position.
Glass-fibre Reinforcement Corrosion: Unlike steel reinforcement, glass fibres are introduced in random orientation and throughout the paste. Typically, as filament bundles (of around 50 filaments) the fibres will be of variable length (up to 2 cm typically). As in the AAR, the highly alkaline cement pore fluid attacks the siliceous glass to produce a gel which imbibes water. The result is loss of effective fibre diameter as the gel continues to form. Occasionally, Ca(OH)2 crystals attach themselves to the fibre and degrade the fibre locally (notching) but the more general attack occurs even when pozzolans are added to minimise notching. This degrading reaction prohibits the use of glass reinforced concrete (GRC) as sole reinforcement in structural concrete.